South India -Shatavahanas, Gangas Class 8 Questions and Answers KSEEB 8th Social Science









You can Download Chapter 2 State Questions and Answers, Notes, 1st PUC Political Science Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
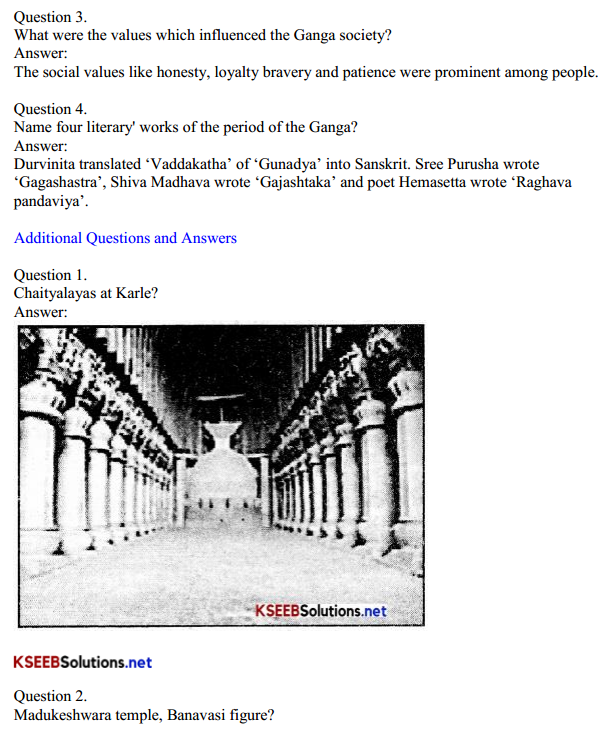
Question 1.
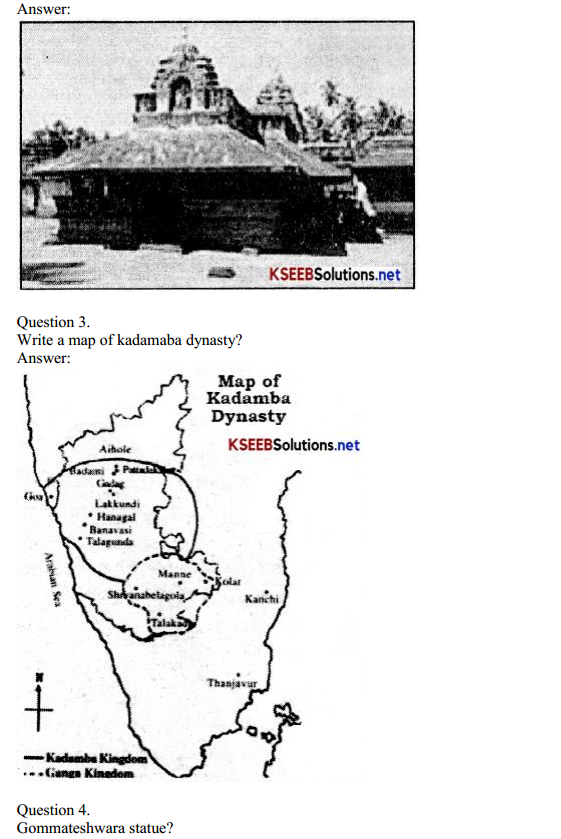
Which is the most essential human organisation?
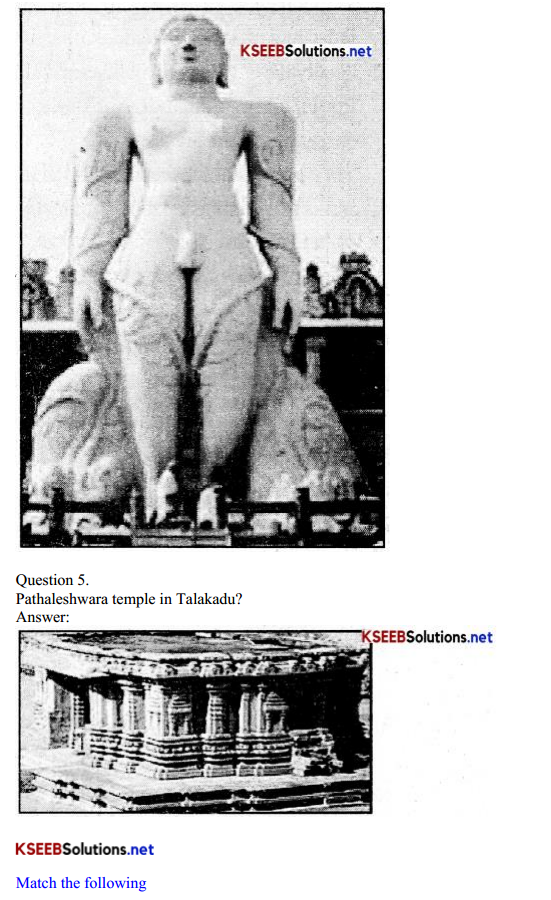
Answer:
State is the most essential human organisation.
Question 2.
What is State?
Answer:
The state is politically organised people in a definite territory.
Question 3.
What is the main purpose of a state?
Answer:
The welfare of the people is the main purpose of a state.
Question 4.
Who said that man is a social and. also a political animal?
Answer:
Aristotle says man is a social and also a political animal.
![]()
Question 5.
What are the root words of the state?
Answer:
Romans says ‘‘civitas’’ and Italians say ‘statio’.
Question 6.
Give an example of a city-state.
Answer:
Sparta and Athens are examples of a city-state.
Question 7.
Who called the state as ‘Civitas’?
Answer:
Romans called the state as ‘Civitas’.
Question 8.
Who introduced the term state for the first time?
Answer:
Italian thinker Machiavelli introduced the word state in Political Science.
Question 9.
Who is the author of “The Prince”?
Answer:
Machiavehi is the author of ‘The prince’.
Question 10.
Which is the supreme of all associations?.
Answer:
The State.
Question 11.
Which association serves people from birth to death?
Answer:
State.
![]()
Question 12.
Who advocated the Sapthanga theory of the state?
Answer:
Kautilya advocates the Sapthanga theory of the state.
Question 13.
According to Plato what should be the population of the state?
Answer:
According to Plato, the population of the state is 5040.
Question 14.
Who said that the population of the state should be 10,000?
Answer:
Rousseau says the population of the state should be 10,000.
Question 15.
Who is the author of the book ‘Arthashastra’?
Answer:
Kautilya is the author of the book ‘Arthashastra’.
Question 16.
Name the state with the highest population in the world.
Answer:
The hight population state in the world is China.
Question 17.
Name the state with the lowest population in the world.
Answer:
The lowest population in the world is Nauru.
![]()
Question 18.
Who said that ‘Good people build a good state’.
Answer:
Aristotle says ‘Good people build a good state’.
Question 19.
What is the territory?
Answer:
A definite land which is essential for a state is called territory.
Question 20.
What is the government?
Answer:
An agent of a state is called the government.
Question 21.
Which is called as an agent of the state?
Answer:
The government is called an agent of the state.
Question 22.
Which organ is called as the ‘Heart and soul’ of the state?
Answer:
The government is called the ‘Heart and soul’ of the state.
Question 23.
What is meant by Sovereignty?
Answer:
Sovereignty is the Supreme Power of the State.
Question 24.
What is an association?
Answer:
A group of people which have a specific objectives is called an association.
Question 25.
Which is called as ‘The association of associations’?
Answer:
State.
![]()
Question 26.
What is society?
Answer:
A web of social relationships is called society.
Question 27.
Who said ‘Society is the web of social relationship’?
Answer:
Max Weber says society is the web of social relationship.
Question 28.
Which is known as the ‘crown association’?
Answer:
State.
Question 29.
What is the root word of the term ‘Nation’?
Answer:
The term ‘Nation has been derived from the Latin word ‘Natio’.
Question 30.
Who said ‘Nation is state + Nationality’?
Answer:
Gilchirst says ‘Nation is state + Nationality’.
Question 31.
When did India became a sovereign state?
Answer:
India becomes sovereign state on 15th August 1947.
Question 32.
What is nation?
Answer:
An emotional feeling of oneness is called a nation.
Question 33.
What is meant by the State?
Answer:
State means an assembly of people occupying a definite territory, having an organized government and subject to no outside control. (Sovereignty)
Question 34.
What is the root word of the term ‘STATE’?
Answer:
The Teutonic term ‘Status’ may be regarded as the root word of the term State in Political Science.
![]()
Question 35.
What are the four Elements of the State?
Answer:
The four Elements of the State are:
Question 36.
Which is the most important element of the State?
Answer:
The most important element of the state is sovereignty.
Question 37.
Mention the two aspects of Sovereignty.
Answer:
Two aspects of Sovereignty are: Internal and External Sovereignty.
Question 38. Which is the most powerful of all Social Institutions.
Answer:
The State.
Question 39.
Which is the root word of Sovereignty?
Answer:
The root word of Sovereignty is the Latin word ‘Super nus’ which means State.
Question 40.
Who wrote the book ‘The Prince’?
Answer:
Machiavelli of Italy.
Question 1.
What is the meaning of state.
Answer:
The state is politically organised people in a definite territory.
Question 2.
Why is the state inevitable?
Answer:
The state is inevitable to ensure the protection of the life and property of the inevitable.
Question 3.
Name any two theories of the origin of the state.
Answer:
Divine theory and social contract theory are the two theories of the state.
Question 4.
Define state.
Answer:
According to Woodrow Wison, the state is people organised for law within a definite territory.
Question 5.
What is the welfare state?
Answer:
A state which provides the basic facilities to all the people from birth to death is called a welfare state.
Question 6.
Mention the organs of Sapthanga Theory.
Answer:
Swami (King), Amatya (Minister), Janapada (Population and territory), Kosha (treasury), Bala (Army), Durga (Fort). Mithra (International relations).
![]()
Question 7.
Mention the four elements of a state.
Answer:
Population, definite territory, sovereignty, and government.
Question 8.
Write any two advantages of a limited population.
Answer:
Question 9.
Name any two states which have a large population.
Answer:
Question 10.
Name any two advantages of small territory.
Answer:
Question 11.
Write any two advantages of large territory.
Answer:
Question 12.
Name any two states with large territory.
Answer:
Question 13.
Name any two states which have small territory.
Answer:
Question 14.
Write the meaning of government.
Answer:
The government his an agent which fulfills the interests of a state.
![]()
Question 15.
Mention the organs of government.
Answer:
Question 16.
Write any two forms of government.
Answer:
Question 17.
What is the meaning of sovereignty?
Answer:
A supreme power of the state over its citizens is called sovereignty.
Question 18.
Name the aspects of sovereignty.
Answer:
Question 19.
What is Internal Sovereignty?
Answer:
A Country that is free overall its individuals and associations within its territory is called internal Sovereignty.
![]()
Question 20.
What is external sovereignty?
Answer:
Freedom of a state which ensures to have an independent foreign policy with other countries is called external sovereignty.
Question 21.
What is the meaning of Society?
Answer:
The term Society refers to the whole complex of the Social relationship of Man. A Society may be defined as an association of individuals.
Question 22.
Write the difference between State and Society.
Answer:
Society was there before the State. It is prior to the State. Society has got variety purposes whereas the State only one object.
Question 23.
What is an associations? Give an example.
Answer:
Associations are a group of people united together to realize some common goals. They are Social institutions. The State is one among associations. Ex. Lions club.
![]()
Question 24.
Mention the two differences between State and Association.
Answer:
Membership of the State is Compulsory. It is Voluntary or Optional in the case of association. A Person can be a Citizen of only one State at a time. On the other hand, he can become a member of many associations at the same time.
Question 25.
Write the meaning of the Nation.
Answer:
An emotional feeling of oneness among the people who belongs to common stock is called a nation.
Question 26.
Write any two differences between the State and Nation.
Answer:
Question 27.
What is the main difference between the State and Government?
Answer:
The government is the agent of the State. It is the agent through which the will of the State is formulated expressed realized. The State is an abstract concept government, is its concrete
form.
Question 28.
Why is the association ‘Voluntary’?
Answer:
Associations are formed to serve different aspects of Social life. Each having its own aims and objectives. Its membership is optional, so they are Voluntary.
![]()
Question 29.
Mention the Elements of Nationality.
Answer:
Common Race, Common Religion, Common Language, Common Culture and Traditions, Common History.
Question 30.
Who is an Alien?
Answer:
An Alien is one who owes his allegiance to his home state and not to the state where he lives temporarily. He enjoys only Civil Rights.
Question 31.
Give the meaning of Secular State.
Answer:
A country that does not have any religion as State religion, and it gives equal importance to all religions and does not insist on any individual to follow a particular religion is called a Secular State.
Question 32.
What is meant by Power?
Answer:
It is one of the subject matters of Political Science. It is the capacity to influence the behavior of others and also the capacity to get things done from others.
Question 33.
What is Dual Citizenship? Give Example?
Answer: The Citizen of a Nation Enjoys two kinds of Citizenship, one of his nation and another is of the state where he actually resides.
E g: U S A and Switzerland.
Question 34.
Name the two features of Power?
Answer:
Question 1.
Explain the Importance of the state.
Answer:
1. To realize social good:
the existence of the state enables the realization of social good.
2. Protection of rights and duties:
rights and duties enjoyed by men today can exist only within a state. It also promotes a smooth relationship between the government and the governed.
3. Protection to life and property:
state is the basic structure for the maintenance of law and order, so the property of the people is also protected by the state.
4. Protection of Weaker section of Society:
The state also protects the weaker section of the society like women, children, and minorities.
5. State creates Order in Society:
A society without a state will be one of anarchy. Political organization is necessary7 to prevent anarchy.
6. State creates order and Progress m the world:
The conception of the state is not only important within a state to create order but even outside the state in the international sphere.
![]()
Question 2.
What is Sovereignty? Explain the two aspects.
Answer:
The supreme power of the state is called Sovereignty. It can not be questioned by any person in the state. The two aspects of Soverignity are as follows.
Internal sovereignty means the supremacy of the state overall individuals and associations within its territory. Every independent state has some person, assembly or group which has the final legal power to command and enforce the authority on behalf of the state. It issues all the orders within its jurisdiction and receives orders from no one. It is absolute and has no legal limitations. Hence the laws of the state are final and binding.
External sovereignity means the state is independent of all external control and force which can coerce a state into obedience. A state is free and equal with other states. An independent state can enter into any treaty or agreement and have its own foreign policy to carry on its own foreign affairs without any interference from other states.
Question 3.
What is the difference between State and Society?
Answer:
|
State |
Society |
| 1. State is supreme institution. | 1. Society is not supreme Institution. |
| 2. State originated after the society. | 2. Society originated prior state. |
| 3. Scope of state is limited. | 3. Scope of society is wider. |
| 4. Definite Territory is essential for the state. | 4. Definite territory is not essential for society. |
| 5. State consists of organized people | 5. Society consists of both organized and unorganized people. |
| 6. State has sovereignty. | 6. Society has no sovereignty. |
| 7. State functions through the government. | 7. Society has no government. |
| 8. State is one of the association in the society. | 8. Society has many associations of which state is one. |
| 9. Membership of state is compulsory. | 9. Membership of society is obligatory. |
| 10. State Studies about Political system. | 10. Society studies about social system. |
| 11. State controls our external relations. | 11. Society influences on our internal relations. |
| 12. Rules of the state are compulsory. | 12. The rules of society are not compulsory. |
| 13.State has the power to punish the people. | 13. Society cannot punish people. |
| 14. State has a legal system. | 14. Society has no legal system. |
![]()
Question 4.
Distinguish between State and Association.
Answer:
|
State |
Voluntary Association |
| 1. Definite territory is essential element of state | 1. Associations have no definite territory. |
| 2. Membership is compulsory man cannot Give up the membership. | 2. Membership is temporary; man can give up the membership. |
| 3. Individual can get the membership of only a state. | 3. Individuals can get the membership of various associations as he pleases. |
| 4. State is permanent and continuous. | 4. Associations are temporary. State can control and abolish them at any time. |
| 5. State has sovereignty. | 5. Associations have no sovereignty. |
| 6. State’s functions are wider. | 6. Functions of associations are narrower. |
Question 5.
Explain the differences between State and Nation.
Answer:
|
State |
Nation |
|
1. State is Political Institution. |
1. Nation is a Social Institution. |
| 2. The State should be a nation. | 2. Nation can become state. |
| 3. The state four elements such as population, territory government, and sovereignty. | 3. Nation has no four elements. It has Population and territory. |
| 4. State expresses the political concept. | 4. Nation has internal, psychological, Spiritual concepts. |
| 5. State is limited. | 5. Nation is universal. |
| 6. State has sovereignty. | 6. Nation has no sovereignty. |
| 7. State may lose its state ship. | 7. Nation is permanent. |
| 8. State is Supreme. | 8. Nation is not supreme. |
| 9. State is organized. | 9. Nation has social, religious, cultural nature. |
| 10. State has the power to punish the people. |
10. Nation has no power to punish the people. |
Question 6.
Explain any two elements of Nationality.
Answer:
Common Race:
[Some writers Say racial purely is a factor, which helps in the formation and continuance of nationality].
People belonging to the same race develop strong bonds of unity and form nationality.
There is no pure race in any nation. The USA, Canada, Switzerland and India are belonging to various origins . Thus the racial unity is a desirable factor for the formation of nationality but an absolutely essential factor.
Common Language:
It brings people together. People who speak common language have a strong bond of relationship and unity. It helps people to communicate with each other. For e. g: People of Switzerland speak their languages, Hench, German and Italian but form one nationality. In India, we speak many languages but we belong to one nationality.
![]()
Question 7.
Write a note on the general duties of a citizen.
Answer:
Duty is an obligation and a responsibility of a citizen. A Citizen of a state is directed to discharge some functions and prohibited from certain functions. These are called duties. They are broadly divided into moral and legal duties. The legal duties are general duties that are enforced and recognized by the laws of the state. They are compulsory. If a citizen neglects his duty he is punished. The important general duties of a citizen are:
1. Loyalty to the state:
Every citizen should be loyal to his state. This includes his duty to defend the state during the war and his involvement in developmental activities, to Protect and promote unity and integrity of the state and for the maintenance of peace, order, and security of the state.
2. Obedience to law:
Every citizen should obey the laws as disobedience leads to confusion
and disorder.
3. Payment of Taxes:
Every citizen must honestly pay and promptly taxes for the economic development of the state.
4. Honest exercise of the right to vote:
This duty enables citizens to elect gout of their choice which works for development, welfare, security, etc.
Question 8.
Explain the Nature of State.
Answer:
Question 1.
Define state and explain its essential elements. .
Answer:
Aristotle defines the state as “A Union of families and villages having for its end a perfect and self-sufficient life, by which we mean a happy and honorable life”.
According to Bluntschli. The state is Politically Organized people According to Wilson, “The state is a people organized for law within a definite territory”. According to Laski, “The state is a territorial society divided into government and subject, etc, claiming within its allotted physical area, a supremacy over all other institutions”.
The above definitions show that there are four essential elements of the state.
1. population:
People are the most important essential of the state. If there are no people there will be nobody to rule and nobody to be ruled. A vacant island or desert can’t constitute a state. Hence, the population is essential for state and without population there can be no state.
For Aristotle, the number should be neither too large nor too small. It should be large enough to be self-sufficient and small enough to be well-governed. The actual population is less than the optimum; the natural resources can’t be fully utilized. If it is more, poverty will be the result.
2. Territory:
Territory is a definite area of earth’s surface where people reside permanently. We can’t think of a state without territory.
The area within these boundaries constitutes the “Territorial Jurisdiction” of a state.
There is no fixed limit to the size of the territory, it may be generally laid down that there should be a proper balance between population and territory.
3. Government:
For the peaceful existence, gout is very much essential for a state. The government is the machinery, which regulates controls and directs human behavior in society. It is an agency of the state through which the will of the state is formulated, expressed and executed. The entire population of a political community makes a state. The gout is composed of few persons duly elected or selected. There are branches of government. They are legislature, executive and judiciary. The form of government differs from state to state.
4. Sovereignty:
It means the supreme power of the state. The term sovereignty derived from the Latin word supreranus, which means supreme. There are two aspects of sovereignty Internal and external ‘sovereignty’. Internal means supreme legal authority and group within its territory. External means freedom of state from any kind of outside control. It means Pot – Independence of state. ‘Sovereignty’ is the most important element of the State.
![]()
Question 2.
Explain the Importance of state.
Ans.
1. To realize social good:
The existence of the state enables the realization of social good.
2. Protection of rights and duties:
Rights and duties enjoyed by men today can exist only within a state. It also promotes a smooth relationship between the government and the governed.
3. Protection to life and property:
State is the basic structure for the maintenance of law and order, so the property of the people is also protected by the state.
4. Protection of Weaker section of Society:
The state also protects the weaker section of society like women, children, and minorities.
5. State creates Order in Society:
A society without a state will be one of anarchy. Political organization is necessary to prevent anarchy.
6. State creates order and Progress in the world:
The conception of the state is not only important within a state to create order but even outside the state in the international sphere.
Question 3.
What is meant by Nation and Nationality?
Ans.
Writers on political science are not agreed on the exact meaning of word nationality. The term. Nation and Nationality have been used interchangeably. Both the term Nation and Nationality are derived from the same root word ‘Nato’, which means born, birth, or race. The word nation has acquired a political meaning whereas nationality acquired spiritual meaning.
According to Lord Bryce, a nation means a self governs nationality.
To Gilchrist, a nation means state plus nationality refers to the feeling of oneness, which exists among a group of people with common race, religion, common language, etc.
The meaning of the word nation is not very clear as we have already stated this word derived from the word ‘Nato’ which means birth.
The Major points of difference are as follower.
Students can Download Maths Chapter 13 Statistics Ex 13.3 Questions and Answers, Notes Pdf, KSEEB Solutions for Class 8 Maths helps you to revise the complete Karnataka State Board Syllabus and score more marks in your examinations.
Question 1.
Runs scored by 10 batsmen in a one-day cricket match are given. Find the average run scored. 23, 54, 08, 94, 60, 18, 29, 44, 05, 86
Answer:
Σ x = 23 + 54 + 08 + 94 + 60 + 18 + 29 + 44 + 05 + 86 = 421
N = 10
Average = Mean =
Question 2.
Find the mean weight form the following table:
| Weight (kg) | 29 | 30 | 31 | 32 | 33 |
| No. of children | 02 | 01 | 04 | 03 | 05 |
Answer:
| Weigh t(kg) | No. of children(x) | ‘ f |
| 29 | 02 | 58 |
| 30 | 01 | 30 |
| 31 | 04. | 124 |
| 32 | 03 | 96 |
| 33 | 05 | 164 |
| N= 15 | Zf = 473 |
Mean = \(\frac{\Sigma f_{X}}{N}=\frac{473}{15}=31.53\)
Question 3.
Calculate the mean for the following frequency distribution.
| Mark | 10-20 | 20-30 | 30-40 | 40-50 | 50-60 | 60-70 | 70-80 |
| Frequency | 3 | 7 | 10 | 6 | 8 | 2 | 4 |
Answer:
| Marks | Frequency | Midpoint | fx |
| 10-20 | 3 | 15 | 45 |
| 20-30 | 7 | 25 | 175 |
| 30-40 | 10 | 35 | 350 |
| 40-50 | 6 | 45 | 270 |
| 50-60 | 8 | 55 | 440 |
| 60-70 | 2 | 65 | 130 |
| 70-80 | 4 | 75 | 300 |
| N = 40 | Efx = 1710 |
Mean = \(\frac{\Sigma f_{x}}{N}=\frac{1710}{40}=42.75\)
Question 4.
Calculate the mean for the following frequency distribution.
| Mark | 15-19 | 20-24 | 25-29 | 30-34 | 35-39 | 40-44 |
| Frequency | 6 | 5 | 9 | 12 | 6 | 2 |
Answer:
| Marks. | Frequency | Midpoint | f(X) |
| 15-19 | 6 | 17 | 102 |
| 20-24 | 5 | 22 | *110 |
| 25-29 | 9 | 27 | 243 |
| 30-34 | 12 | 32 | 384 |
| 35-39 | 6 | 37 | 222 |
| 40-44 | 2 | 42 | 84 |
| N = 40 | Efx = 1145 |
Mean = \(\overline{X}=\frac{\Sigma f_{X}}{N}=\frac{1145}{40}=28.625\)
Question 5.
Find the median of the data 15,22, 9,20, 6,18,11,25,14.
Answer:
6, 9, 11, 14,(15), 18, 20, 22,25 (Ascending order)
N = 9,
Median = 15.
Question 6.
Find the median of the data 22,28,34, 49, 44, 57,18,10,33, 41, 66, 59.
Answer:
10, 18, 22, 28, 33, 34, 41, 44, 49, 57, 59, 66 (Ascending order)
N = 10
∴ Median =
Question 7.
Find the median for the following frequency distribution table.
| Class interval | 110-119 | 120-129 | 130-139 | 140-149 | 150-159 | 160-169 |
| Frequency | 6 | 8 | 15 | 10 | 6 | 5 |
Answer:
| Class interval | Frequency (f) | Cumulative frequency (fc) |
| 110-119 | 6 | 6 |
| 120-129 | 8 | 14 |
| 130-139 | 15 | 29 |
| 140-149 | 10 | 45 |
| 160-169 | 5 | 50 |
| N = 50 |
N = 50
\(\frac{\mathrm{N}}{2}=\frac{50^{25}}{\not 2}=25\)
∴ Median class is 130 – 139
LRL = 129.5
Fc = 14
Fm = 15
i = 10
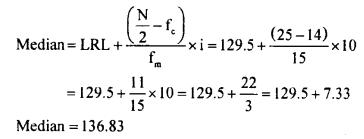
Question 8.
Find the median for the following frequency distribution table.
| Class interval | 0-5 | 5-10 | 10-15 | 15-20 | 20-25 | 25-30 |
| Frequency | 5 | 3 | 9 | 10 | 8 | 5 |
Answer:
| Class interval | Frequency (t) | Cumulative frequency (fc) |
| 0-5 | 5 | 5 |
| 5-10 | 3 | 8 |
| 10-15 | 9 , | 17 |
| 15-20 | 10 | 27 |
| 20-25 | 8 | 35 |
| 25-30 | 5 | 40 |
| N = 40 |
N = 40
\(\frac{N}{2}=\frac{40}{2}=20\)
∴ Median class is 15 – 20
LRL = 15
Fc = 17
Fm = 9
i = 5
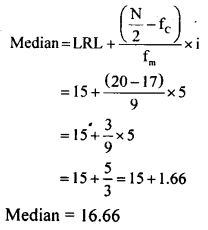
Question 9.
Find the mode for the following data.
(i) 4,3,1,5,3, 7, 9,6 Answer: Mode = 3
(ii) 22,36,18,22,20,34,22, 42, 46,42
Answer:
Mode = 22
Question 10.
Find the mode for the following data
| X | 5 | 10 | 12 | 15 | 20 | 30 | 40 |
| f | 4 | 8 | 11 | 13 | 16 | 12 | 9 |
Answer:
Mode = 20 (It has the highest frequency)
You can Download Chapter 1 Making of Indian Society and Demography Questions and Answers, Notes, 2nd PUC Sociology Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
Question 1.
How is the term Demography derived?
Answer:
The term Demography is derived from two Greek words i.e. demos (people) and graphein (describe), implying the description of people.
Question 2.
What is Demography?
Answer:
Demography is the systematic study of population.
Question 3.
Name any one type of Demography.
Answer:
Formal Demography.
Question 4.
Give one major characteristic of Demographic profile of India.
Answer:
Size and Growth of India’s Population.
Question 5.
Mention sex ratio of India according to 2011 census.
Answer:
940.
![]()
Question 6.
Which district in Karnataka is Selected to Implement Beti Padavo and Beti Bachavo Programme?
Answer:
Vijyaypura District.
Question 7.
Name one racial group of India.
Answer:
Negritos.
Question 8.
Give one sub division of Mongoloid race in India;
Answer:
Paleo Mangoloid.
Question 9.
Who introduced Christianity to India?
Answer:
St Thomas and St Bharathaomew.
Question 10.
What is unity?
Answer:
Unity implies oneness, or a sense of we-ness.
Question 11.
Name one basis of diversity in India.
Answer:
Linguistic Diversity.
Question 12.
Name one basis of unity of India.
Answer: Religious unity.
Question 13.
What is National Integration?
Answer:
National integration refers to national unity and a sense of belonging to the nation.
Question 14.
Name anyone challenge to National Integration.
Answer:
Communalism.
![]()
Question 15.
Name any one religious community of India.
Answer: Hindus.
Question 16.
Name any one ancient name of India.
Answer:
Bharathakanda.
Question 17.
What does DEMARU stands for?
Answer:
Data from 2001 census to coin another acronym – DEMARU – where D stands for daughter and MARU stands for killing. In English “E” will denote elimination. Punjab, Hariyana, Himachal Pradesh & Gujarath, Maharashtra as DEMARU states, where the sharp decline in the Juvenile sex ratio.
Question 18.
Which European colonial group first entered India?
Answer:
Portuguese.
Question 19.
Expand the UNPF
Answer:
United Nations Population Fund.
Question 20.
Expand the Abrivation BIMARU
Answer:
Bihar, Madhya Pradesh, Rajasthan and Uttar Pradesh.
Question 21.
Define formal Demography.
Answer:
Formal demography is primarily concerned with the measurement and analysis of the components of population change. Its focus is on quantitative analysis for which it has a highly developed mathematical methodology suitable for forecasting population growth and changes in the composition of population.
Question 22.
Define social Demography.
Answer:
Social demography, on the other hand, enquires into the wider causes and consequences of population structures and change. Social demographers believe that social processes and structures regulate demographic processes; like sociologists, they seek to trace the social reasons that account for population trends.
Question 23.
In which year first census was conducted.
Answer:
1867-72.
Question 24.
Which is the oldest civilization of India.
Answer:
Indus valley civilization.
Question 25.
In which was year Kannada language has declared as one of the classical languages.
Answer:
2008.
Question 26.
What is the total population of Karnataka according to 2011 census?
Answer:
6,10,95,297.
Question 27.
What is the sex ratio of Karnataka according to 2011 census?
Answer:
973.
![]()
Question 28.
Which is the most populated district in Karnataka.
Answer:
Bangalore District.
Question 29.
Which district has Highest Sex Ratio in Karnataka? ‘
Answer:
Udupi District (1094).
Question 30.
Which district has Lowest Sex Ratio in Karnataka?
Answer:
Bangalore District (916).
Question 31.
What is the density of Karnataka according to 2011 census?
Answer:
319.
Question 32.
Which district in Karnataka registers Highest density?
Answer:
Bangalore District (2985).
Question 33.
Which district in Karnataka registers Lowest density?
Answer:
Kodagu District (134).
Question 34.
Which district has recorded highest SC population in Karnataka?
Answer:
Kolar District.
Question 35.
Which district has recorded highest ST population in Karnataka?
Answer:
Raichur District.
Question 36.
Which district in Karnataka has retained highest literacy rate?
Answer:
Dhakshina Kannada (88.57).
Question 37.
Which district in Karnataka has retained lowest literacy rate?
Answer:
Yadagiri(51.83).
Question 38.
What is the total literacy rate of Karnataka in 2011?
Answer:
75.39%.
Question 39.
What is Sex Ratio?
Answer:
Number of females for 100 male population.
Question 40.
State a Reason for Imbalance in Sex Ratio.
Answer:
Sex selective abortion.
Question 41.
Which year is considered as demographic divide?
Answer:
1921.
Question 42.
How many languages were recognized by Indian constitution as official languages?
Answer:
22.
Question 43.
Mention any one Indo-Aryan language
Answer:
Hindu.
Question 44.
Mention any one Dravidian Language.
Answer:
Kannada
Question 45.
Mention any one Austric Language
Answer:
Mundari.
![]()
Question 46.
In which year Government of India declared Kannada as one of the classical language?
Answer:
2008.
Question 47.
Which is the oldest water dispute in the world?
Answer:
Cauvery water dispute.
Question 1.
Define Demography.
Answer:
Demography studies the trends and processes associated with population including – changes in population size; patterns of births, deaths, and migration; and the structure and composition of the population, such as the relative proportions of women, men and different age groups.
Question 2.
Give two major characteristics of Indian Demographic profile.
Answer:
Question 3.
Mention any two factors responsible for decline of child sex ratio.
Answer:
Sex selective Abortions, Neglect of Girls in an Infancy.
Question 4.
Mention any two racial groups of India.
Answer:
Negritos and Mongoloids
Question 5.
Name two difficulties to the process of Aryanzation.
Answer:
![]()
Question 6.
What does DEMARU stands for?
Answer:
Data from 2001 census to coin another acronym – DEMARU – where D stands for daughter and MARU stands for killing. In English “E” will denote elimination. Punjab, Hariyana, Himachal Pradesh & Gujarath, Maharashtra as DEMARU states, where the sharp decline in the Juvenile sex ratio.
Question 7.
Define national Integration.
Answer:
National integration refers to national unity and a sense of belonging to the nation.
Question 8.
What is Regionalism?
Answer: Regionalism refers to an extreme loyalty or love to a particular region which may undermine the interest of the nation.
Question 9.
What is communalism?
Answer:
Communalism is the antagonism practiced by the members of one community against the people of community and religion.
Question 10.
What is Iinguism?
Answer:
Linguism implies one-sided love and admiration towards one’s language and a prejudice and hatred towards others’ languages.
Question 11.
Give two measures to strengthen national integration.
Answer:
Question 12.
State any two dravidian language.
Answer:
Tamil and Kannada.
Question 13.
State any two Indo – Aryan languages.
Answer:
Sanskrit and Hindu.
Question 14.
State any two Austric languages.
Answer:
Mundari and Santhali.
Question 15.
State any two Tibeto – Burman languages.
Answer:
Bodo and Ladaki.
Question 16.
Mention any two classical languages of India.
Answer:
Sanskrit and Tamil.
![]()
Question 17.
Mention any two threats to national Integration.
Answer:
Communalism and Regionalism.
Question 18.
State any two forms of regionalism.
Answer:
Question 19.
Why India is regarded as museum of languages?
Answer:
India is a land of many languages and it has been called as a ‘museum of languages’.
Question 20.
What is demographic Dividend?
Answer:
Demographic dividend refers to demographic or population advantage which is obtained due to numerical domination of the young people in the population. It is an advantage due to less dependency ratio.
Question 21.
What is dependency ratio?
Answer:
Dependency ratio means children less than 14 years and people above 65 years are considered as to be dependent on the rest of the population. In simple terms the ratio of the combined age group 0-14 years plus 65 years & above to the 15-65 years age group is referred to as the total dependency ratio.
Question 1.
Explain the racial groups classified by B.S. Guha.
Answer:
B.S. Guha who identified six major racial elements in the population of India:
In the south, the Kadar, the Irula, and the Paniyan, and in the Andaman Islands, the Onge and Jarwas of the Andamanese have definite Negrito characteristics. Some traits of this group are found among the Angami Naga and the Bagadi of the Rajmahal hills. On the western coast there are some groups with pronounced Negrito traits, but they perhaps represent later arrivals, who came to India with the Arab traders.
The Proto-Australoid group is numerically more significant; most of the tribes of middle India belong to it. These were the people described by the Indo-Aryans as Anas, Dasa, Dasyu, and Nishad – all derogatory terms. The Mongoloid group is sub-divided into two branches – Paleo- Mongoloid and Tibeto-Mongoloid. Tribal groups in the Himalayan region and those in the north-east are of Mongoloid stock. Some Mongoloid features are seen in the non-tribal population of the eastern States – Assam, West Bengal, Manipur, and Tripura.
The Western Brachycephals (sub-divided into the Alpinoid, Dinaric, and Armenoid groups), Alpinoid and Dinaric characteristics are seen in some groups of northern and western India; the Parsis belong to the Armenoid section. The Mediterraneans are associated with the Dravidian languages and cultures. The Nordics were the last major ethnic element to arrive in India and make a profound impact on its culture and society. But before they came a unique civilization had slowly developed in India. It is known as the Indus Valley Civilization.
![]()
Question 2.
Write note on the processes of Aryanization.
Answer:
They were essentially a pastoral people with a flair for poetry, philosophical speculation, and elaborate rituals. They regarded themselves as superior and tended to look down upon the earlier inhabitants of the land, for whom they coined several derogatory terms. They were required to marry within their own group, i.e., practise endogamy, and had some elementary notions of ritual purity and pollution which governed their physical contacts and commensal – inter-dining – relations with others. This led to the origin of the Vamas and also of Jati (caste). Commensality and sexual relations within different tribal groups and earlier ethnic groups were governed by customary norms and taboos, and the advent of the Indo-Aryans brought in refinements and complexities in them.
The Indo-Aryans were divided into three groups – the Rajanya (warriors and the aristocracy), the Brahmana (priests), and the Vaishya (cultivators). These were Dvija (twice-born) groups
born first at physical birth and a second time when initiated into Varna status. The Shudras were the fourth Vama; they were from outside the Indo-Aryan group and were perhaps the progeny of unions between the Indo-Aryan and the Dasa (the pre-Aryan .inhabitants of the land). They emerged as cultivators, but were denied twice – bom status. Outside the four-fold vertical Vama structure, there was a fifth group – Avarna or the Pancham – whose ethnic status was so low and their occupations so degraded and polluting that any physical contact with them was prohibited for the twice-born and the Shudra.
The process of Aryanization of the sub-continental traditions was neither smooth nor complete. Its earlier phase was characterized by considerable cultural conflict and warfare. Much Accommodation and Compromise were also taking place.
This necessitated greater harmony with the older inhabitants; in any case, some kind of synthesis between them was already taking place. As a result the non-Aryans adopted some elements of Indo-Aryan ritual and their philosophy of social organization, while retaining their own ethnic and regional identities. Pluralism was being stabilized and a cultural mosaic being formed.
Question 3.
Explain the nature of diversities in India.
Answer:
The term Diversity denoting collective differences so as to find out dissimilarities among the people: geographical, religious, linguistic etc. Thus all these differences presuppose collective differences or prevalence of variety of groups and culture. Indian society is characterized by unity as well as diversity.
The Nature of Diversities in India
Primarily there are major four types of diversities in India, which are;
(1) Regional Diversities: It is evident that there are extreme regional diversities in its geographic features. Indian territory contains huge mountains, thick forests, numerous rivers and etc. It is only in India that there are different regions, different types of climates and the temperatures. India is a vast country. From the Himalayas in the North to Indian Ocean in the south. There are difference in altitude, temperature, Flora and Fauna. India has every conceivable type of climate, temperature and physical configuration. There is the scorching heat of Rajastan and the biting cold of the Himalayas, Rainfall varies from 1200 to 7.5 cms per year. The result is that India has some of the wettest and driest areas in the world. India also possesses arid desserts and fertile riverine lands, bare and hilly tracts and luxuriant open plain.
(2) Linguistic Diversities: Language is another source of diversity. It contributes to collective identities and even to conflicts. The Indian Constitution has recognized 22 languages in the 8th schedule for its official purposes but as many as 1652 languages and dialects are
spoken in the country. According to Grierson’s Linguistic Survey of India, these languages belong to five linguistic families, namely; Indo-Aryan languages, Dravidian languages, Austric languages, Tibeto – Burman languages and European languages.
This makes language planning and promotion difficult. But the mother tongue does evoke strong sentiments and reactions. As a consequence of this multiplicity, there is considerable bilingualism and administration has to use more than one language. Linguistic diversity has posed administrative and political challenges.
(3) Religious Diversities: There are eight major religious communities in India. Hindus constitute 82.7%, Muslims 11.8%, Christians 2.6%, Sikhs 2%, Buddhists 0.7%, Jains 0. 4%, Zoroastrians 0.3%, and Jews 0.1 %. Each major religion is subdivided along the lines of religious documents, sects, and cults. The Hindus are now broadly divided into Shaivite (worshippers of Shiva), Vaishnavaite (worshippers of Vishnu and his incarnations), Shakta (worshippers of the Mother Goddess in various manifestations. Even among them there are sub – divisions based on doctrinal and ritual differences.
Buddhism was spread widely in India once, it lost its hold in the country of its birth and remained confined only to a few pockets. Jainism too, once held wide sway in India, and though its followers are now numerically small, they are found in both the northern and southern States. They have two main divisions: Digamber-unclothed, and Shwetamber. The Indian Muslims are divided broadly into the Sunni and Shia communities.
Indian Christians are divided into Roman Catholics and Protestants and into many denominational churches. Sikhism is synthesizing religion that emphasizes egalitarianism. Grantha Sahib is the holy book and Amritsar Golden Temple is one of their holy piligrimage. The Parsis are a small community, but they have played an important role in India’s industrial development. The Jewish has been established in India for over a millennium. They also had White and Black divisions and prohibition on inter-marriage and inter-dining, but they all worshipped in the same synagogues. The Jati-like restrictions are much less in Bombay and Cochin, to where many Jews have migrated.
(4) Cultural and Ethnic Diversities: Another important source of diversity is the cultural diversity. The people differ considerably in their social habits. Cultural difference varies from state to state. The conflicting and varying shades of blood, strains, culture, and modes of life, the character, conduct, beliefs, morals, food, dress, manners, social norms, Socio-Religious customs, rituals and etc. causes cultural and ethnic diversities in the country. Dr. R.K. Mukherji rightly said that “India is a museum of cults and customs, creeds and culture, faiths and tongues, racial types and social systems”. Another important source of diversity is the cultural identity of particular communities and region.
![]()
Question 4.
Explain the unity in India.
Answer:
In India aspects of Diversity is as follows as :
(1) Regional Unity: The Natural boundaries provide India a. geographical unity. In ancient times India was known as Bharatavarsha, Bharathakanda, Jambudweepa. This symbolizes the significance of historical unity. The very name “Bharatavarsha” has occupied an important place in the minds of poets, political philosophers, and religious thinkers. Each of them has conceived of the country as a single expanse from the Himalayas to Kanyakumari, a country ruled by one king Bharatha. The concept of Mother India also indicates the realization of geographical unity.
(2) Linguistic Unity: Despite the presence of number of languages, India also possesses lingual unity. Sanskrit as a common base of Indian languages provides the basis of unity as a result of which the linguistic multiplicity has been solved. Simultaneously Sanskrit became the language of Hindu culture and all classics were composed in this language, which demanded reverence and respect. People may speak different languages in different regions but they have common language of Engl ish and Hindi to communicate with each other. The formation of linguistic states and using regional languages as medium of teaching at schools, colleges and universities are the products of Independence.
In 2004 the govt, of India declared that languages that met certain requirements could be accorded the status of a classical language in India. Tamil (2004), Sanskrit (2005), Kannada (2008), Telugu (2008), Malayalam (2013) and Oriya (2014) are declared as classical languages of India. Thus it is an effort to restore linguistic heritage of India.
(3) Religious Unity: In spite of the religious diversities, it possesses religious unity. The feelings of each religious groups are the same, each accepts the truth of immortality of > soul, temporary nature of world, belief in rebirth, the doctrine of karma, Salvation, Contemplation etc., There may be differences in the way these elements are treated but each religion preaches a fundamentally single religious faith and shares a belief in purity ; and values of life in respect of belief in unseen power, benevolence, piety, honesty and liberality, with every religious faith. The worshippers may visit different centres of pilgrimage, but all have a common goal of “Earning religious merit by visiting a sacred place”. India is the sacred land not only for the Hindus but also for Sikhs, Jains and Buddhists. The Muslims and Christians too have several sacred centres of pilgrimage in India.
(4) Cultural Unity: In art and architecture, dress and food, literature, music and dance, sports and cinema, medicine and technology there was a fusion of style and the emergence
of new forms which were the result of their combined efforts. Thus it became apparently clear from the above account that running through various diversities. India has been helped both by nature and nurture, by her geographical condition and historical experiences, by her religious ethics, and political ideas. To realize a unity to perceive, preserve and strengthen the thread of basic unity which makes India a fine example of unity in diversity, transcending birth, caste, language, ethnicity and religious groupings to establish a big society and a big nation.
Modem education, the development of a network of transport and communications, industrialization and urbanization provided new bases for unity.
![]()
Question 5.
Discuss briefly the challenges to National Integration.
Answer:
There are many challenges to national integration. They are as follows:
(1) Regionalism: Regionalism is expressed in the desire of people of one region to promote their own regional interest at the expense of the interests of other regions. It has often led to separatism and instigated separatist activities and violent movements. It has also gained tremendous momentum during the recent years because it is exploited by the selfish politicians Thus, regionalism has challenged the primacy of the nationalistic interests and undermines national unity. Regionalism assumed mainly four forms.
(2) Communalism: Communalism is the antagonism practiced by the members of one community against the people of community and religion. Bipin Chandra holds that communalism is the product of a particular society-, economy and polity, which creates problems. Asghar Ali Engineer, Moin Shakir and Abdul Ahmed try to explain communalism as an ideological tool for propagation of economic and political interests. According to them, it is an instrument in the hands of the upper class to concentrate power by dividing people. The elites strive to maintain a status quo against transformation by dividing people on communal and religious lines.
(3) Linguism: Linguism implies one-sided love and admiration towards one’s language and a prejudice and hatred towards others’ languages. India is a land of many languages and it has been called as a ‘museum of languages’. Diversity of languages has also led to linguism. It has often been manifested into violent movements posing threat to national integration. Linguistic tensions are prevailing in the border areas which are bilingual.
(4) Extremism and Terrorism: Extremism and terrorism have emerged during the recent years as the most formidable challenges to national integration. Extremism refers to the readiness on the part of an individual or group to go to any extreme even to resort to undemocratic, violent and harmful means to fulfill one’s objectives. In the past India has been facing the problems of terrorism since independence. India has faced this problem in Nagaland (1951), Mizoram (1966), Manipur (1976), Tripura (1980) and West Bengal in (1986).
Terrorism in India is essentially the creation of politics. According to According to Prof. Rama Ahuja there are four types of terrorism India, (1) Khalistan oriented terrorism in Puniab (2) Militants terrorism in Kashmir. (3) Naxalite terrorism in west Bengal, Bihar, Madhya Pradesh, Orissa, Andhra Pradesh Telangana, Maharastra, Uttarapradesh Jharkhand, chattisghad out of 318 district 77 districts are highly Naxal poore districts causing lot of bloodshed in these areas. (4) ULFA terrorism in Assam.
The Khalistan oriented Sikh terrorism was based on a dream of theocratic state, Kashmir militants are based on their separate identity. The Naxalite terrorism is based on class enmity. Terrorism in North Eastern India is based on the identity crisis and the grievance situation. In addition to these factors, corruption, poverty, unemployment/youth unrest, widening gap between rich and poor, which are also the major challenges for national integration.
![]()
Question 6.
Briefly explain the Dr. Sampurnananda committee recommendations to strengthen National Integration.
Answer:
The Central Education Ministry organized a ‘Committee for National Integration’ in 1961 under the Chairmanship of Dr. Sampurnanand. The Integration Committee gave some recommendations to promote and strengthen national integration. Some of them are stated below:
Apart from the governmental efforts to achieve the goal of national unity various stakeholders such as educational institutions, religious/cultural associations and mass media should involve in chalking out action-based programmes to enhance awareness/dissemination of traditional values among the masses and increase cultural exchange banking on the richness of our cultural heritage and diversity. Special steps should be taken by various interest groups to speed up development of economically and socially backward groups who are the easy victims of violent activities.
Question 7.
Write a note on BIMARU v/s DEMARU.
Answer:
The results of the 2001 census fully validate the diagnosis of India’s population problem in terms of the dominance of BIMARU States (Bihar, MP, Rajastan, UP) accounted for 39% of India’s population, 42% of growth and 48% of the total Illiterate population and the adverse role of these BIMARU states. To take note of the alarming data from 2001 census to coin another acronym – DEMARU – where D stands for daughter and MARU stands for killing. In English “E” will denote elimination. Punjab, Hariyana, Himachal pradesh & Gujarath, Maharashtra as DEMARU states, where the sharp decline in the Juvenile sex ratio.
These states are in the perverse practice of Foeticide. The unholy alliance between Tradition (Son complex) and Technology (Ultra sound etc.) is playing havoc with Indian society pre-birth sex determination tests & sex selective abortion are rampant in these states. During the last decade in Punjab, the sex ratio (0-6 age group) declined from 875 to 793. In Hariyana the sex ratio (0-6 age group) decreased from 879 to 820.
In Maharashtra every single district showed a decline in the Juvenile sex ratio between 1991-2001. In Himachal pradesh from 951 to 845. In Gujarath from 928 to 878. In Chandigarh from 899 to 845 and in Delhi from 915 to 865. Even though an increase in the sex ratio for the total population was 927 in 1991 to 933 to 2001. In contrast the sex ratio of child population (0-6 age group) which was 945 in 1991 decreased to 927 in 2001. In short the girl child (below 6 years) has lost out badly.
The state-level child sex ratios offer even greater cause for worry. As many as six states and union territories have a child sex ratio of under 900 females per 1000 males. Punjab is the worst off with an incredibly low child sex ratio of 793 (the only state below 800), followed by Haryana, Chandigarh, Delhi, Gujarat and Himachal Pradesh. Uttaranchal, Rajasthan, Uttar Pradesh and Maharashtra are all under 925, while Madhya Pradesh, Goa, Jammu and Kashmir, Bihar, Tamil Nadu, Karnataka and Orissa are above the national average of 927 but below the 950 mark. Even Kerala, the state with the best overall sex ratio does not do too well at 963, while the highest child sex ratio of986 is found in Sikkim.
![]()
Question 8.
What is Demographic Dividend? How it can be utilized?
Answer:
Demographic dividend refers to demographic or population advantage which is obtained due to numerical domination of the young people in the population. It is an advantage due to less dependency ratio. Dependency ratio means children less than 14 years and people above 65 years are considered as to be dependent on the rest of the population. In simple terms the ratio of the combined age group 0-14 years plus 65 years & above to the 15-65 years age group is referred to as the total dependency ratio.
The younger age groups in the age structure is believed to be an advantage for India. Like the East Asian economies in the past decade and countries like Ireland today, India is supposed to be benefitting from a ‘demographic dividend’. This dividend arises from the fact that the current generation of working-age people is a relatively large one, and it has only a relatively small preceding generation of old people to support. But there is nothing automatic about this advantage – it needs to be consciously utilised in the following ways.
a. The demographic advantage or ‘dividend’ to be derived from the age structure of the population is due to the fact that India is one of the youngest countries in the world. In 2020, the average Indian will be only 29 years old, compared with an average age of 37 in China and the United States, 45 in Western Europe, and 48 in Japan. This implies a large and growing labour force, which can deliver unexpected benefits in terms of growth and prosperity.
b. But this potential can be converted into actual growth only if the rise in the working age group is accompanied by increasing levels of education and employment.
c. India is indeed facing a window of opportunity created by the demographic dividend. The effect of demographic trends on the dependency ratio defined in terms of age groups is quite visible. The total dependency ratio fell from 79 in 1970 to 64 in 2005. But the process is likely to extend well into this century with the age-based dependency ratio projected to fall to 48 in 2025 because of continued fall in the proportion of children and then rise to 50 by 2050 because of an increase in the proportion of the aged.
d. This suggests that the advantage offered by a young labour force is not being exploited. Unless a way forward is found, we may miss out on the potential benefits that the country’s changing age structure temporarily offers.
Discuss the Manifestation of Regionalism.
Regionalism assumed mainly four forms:
(i) The Demand for Separate Nation from the Indian Union: The first and most challenging form of Regionalism took was the demand of the people of certain state to succeed from the Indian union and became an independent sovereign states. The first such example was the Tamil community of the state of Madras. The campaign throughout Madras state for the separation of Madras from India and for making it an independent sovereign state of dravidastan. In a similar way Sikhs of Punjab demanded for a separate nation called Khalistan and liberation of Jammu and Kashmir form the Indian union, Insurgency and Secessionist movement in North-Eastern States (Assam, Nagaland, Tripura, Manipur, Meghalaya, Arunachal Pradesh and Mizoram) etc. demand for separate nation.
(ii) Demand for a Separate Statehood: A new form of regionalism has manifested in recent years was the demand for separate statehood in several states of Indian Union, like; Vidarbha states (Maharastra), Telangana, (Andhra Pradesh), Bundelkhand (M.P. and U.P.), Vindhya, Bhojpur and etc. In early 1980s the Jharkhand movement grow much more miltant and the various groups demanding the formation of separate state of Jharkhand, as well in Darjaling, district of West Bengal for Gorkha Land.
(iii) Demand for Full Fledged Statehood: Yet another manifestation of regionalism in India is demand of union territories for full fledged statehood, e.g. Delhi.
(iv) Inter – State Disputes: The first such dispute took place between Karnataka and Maharashtra, Punjab and Haryana. In addition to the above the main disputes are over the sharing of river waters. Over the water of Narmada, Krishna, Kaveri, Bheema and etc. Kaveri water dispute is the oldest water dispute in the world, causing animosity between Karnataka and Tamilnadu frequently.
![]()
Question 1.
Define Demography and Explain the major characteristics of Demographic profile of India.
Answer:
Demography is the systematic study of population. The term Demography is derived from two Greek words i.e. demos (people) and graphein (describe), implying the description of people. The term Demography was coined by Achille Guillard in 1855. Demography studies the trends and processes associated with population including – changes in population size; patterns of births, deaths, and migration; and the structure and composition of the population, such as the relative proportions of women, men and different age groups.
There are different varieties of demography, including Formal demography which is a largely quantitative field, and Social demography which focuses on the social, economic or political aspects of population. All demographic studies are based on processes of counting or enumeration – such as the census or the survey – which involve the systematic collection of data on the people residing within a specified territory.
The American census of 1790 was probably the first modern census, and the practice was soon taken up in Europe as well in the early 1800s. In India, census was conducted by the British Indian government between 1867-72, and regular ten yearly (decennial) censuses have been conducted since 1881. Independent India continued the practice, and seven decennial censuses have been conducted since 1951, the most recent being in 2011. Demographic data are important for the planning and implementation of state policies, especially those for economic development and general public welfare.
The Major Characteristics of the Demographic Profile of India:
(1) Size and Growth of India’s Population: India is the second most populous country in the world after China. According to 2011 census India’s population is 121 crores (1.21 billion). Between 1901-1951 the average annual growth rate did not exceed 1.33%, a modest rate of growth. In fact between 1911 and 1921 there was a negative rate of growth of-0.03%. This was because of the influenza epidemic during 1918-19 : 5%of the total population of the country. The growth rate of population substantially increased after independence from British rule going up to 2.2% during 1961-1981. Since then although the annual growth rate has decreased it remains one of the highest in the developing world.
(2) Age Structure of the Indian Population: India has a very young population – that is, the majority of Indians tend to be young, compare to most other countries, the share of the less than 15 age group in the total population has come down from its highest level of 42% in 1971 to 29% in 2011. The share of the 15-60 age group has increased slightly from 53% to 63%, while the share of the 60+ age group is very small but it has begun to increase (from 5% to 8%) over the same period. But the age composition of the Indian population is expected to change significantly in the next two decades. 0-14 age group will reduce its share by about 11% (from 34% in 2001 to 23% in 2026) while the 60 plus age group will increase its share by about 5% (from 8% in 2001 to about 12% in 2026).
(3) The Declining Sex-Ratio in India: The sex ratio is an important indicator of gender balance in the population. The sex ratio is defined as the number of females per 1000 males. The trends of the last four decades have been particularly worrying – from 941 in 1961 the sex ratio had fallen to an all time low of 927 in 1991 before posting a modest increase in 2001.
According to the Census of India 2011 sex ratio has been increased and now it is 940 females per 1000 males. But what has really alarmed demographers, policy makers, social activists and concerned citizens is the drastic fall in the child sex ratio. The sex ratio for the 0 – 6 years age group (known as the juvenile or child sex ratio) has generally been substantially higher than the overall sex ratio for all age groups, but it has been falling very sharply. In fact the decade 1991-2001 represents an anomaly in that the overall sex ratio has posted its highest ever increase of 6 points from the all time low of 927 to 933, but the child sex ratio in 2011 census has dropped from 927 to 914, a plunge of 13 points taking it below the overall sex ratio for the first time.
(4) Increasing Literacy Rate of Indian Population:. Literacy varies considerably across gender, regions, and social groups. As can be seen from Table No. 4, the literacy rate for women is almost 22% less than the literacy rate for men. However, female literacy has been rising faster than male literacy, partly because it started from relatively low levels. Female literacy rose by about 11.2 percent between 2001 and 2011 compared to the rise in male literacy of 6.2 percent in the same period.
Female literacy was 8.9% in 1951 has increased to 65.4 in 2011 male literacy in the same period wan 27.2% has increased to 82.17. In 1951 total literacy rates 18.3% has increased . to 74.04 in 2011.
(5) Increasing Rural-Urban Differences: According to 2011 Census, 68.8% population lives in rural areas while 31.2% people live in urban areas. The urban population has been increasing its share steadily, from about 17.3% in 1951 to 31.2 in 2011, an increase of about two-and-a-half times.
![]()
Question 2.
Explain the development of Christianity and Islam in India.
Answer:
To understand the texture of Indian society we have also to take note of the long presence of Christianity and Islam in the country. Both were influenced by the prevailing ethos and both made some impact on society. Christianity and Islam acquired some special characteristics in the Indian setting. St. Thomas and St. Bartholomew (A.D. 50) are believed to have brought Christianity to India in A.D. 50. When the Portuguese arrived in India, Christianity was found to have spread over seventeen kingdoms of Kerala. It is believed that Kalyan, near Bombay, emerged as a major centre of Christianity. Apart from St. Bartholomew, a specially invited Stoic philosopher – Pantaenus – was preaching at Kalyan.
The early Christians, however, were held in high esteem by the Hindus. Things began to change with the arrival and establishment of the Portuguese in India. When St. Francis Xavier landed in Goa in 1542, it had become a Christian settlement with fourteen churches and over a hundred clergymen. The Italian Jesuit, Roberto de Nobili, who landed in Goa in 1605 and died in Mylapore (Madras) in 1656, communicated with the people in Tamil and other regional dialects. His discussions with the Brahmans were in Sanskrit. De Nobili was keen to get convinced converts from the top of Hindu society to facilitate the rapid spread of Christianity. He avoided contact with Christians of lower caste origins, including their priests. By the end of the seventeenth century De Nobili and his associates had made many converts.
The later development of Christianity in India is better known because of the association of the Church with foreign powers – The Portuguese, Dutch, British, and French. Considerable evangelical and humanitarian work was done by Christian missionaries of diverse nationalities and denominations. Besides Kerala and Tamil Nadu, there are pockets of Christianity in most States of the Indian Union. At least three of the tribal north-eastern States have substantial Christian majorities. Christians have a sizeable presence among the tribals of Chotanagpur too.
Islam first came to India by peaceful methods, often with the encouragement of Hindu rulers. On the western coast, the Balhara dynasty in the north and the Zamorin of the Malabar coast welcomed Muslim traders and encouraged them to settle in places like Anhilwara, Calicut, and Quilon. They could freely build mosques and practice their religion. Arab and Persian immigrants settled down along the coast and married non – Muslim women. This is how the Nawait (Natia) community of Konkan and the Mappilla (Moplah) community of the Malabar coast emerged. The Labbais, on the east coast of Tamil Nadu, are said to have originated from the union of Tamil women with Arabs who were either shipwrecked or exiled from Iraq.
In the thirteenth and fourteenth centuries several missionaries were active in the Punjab, Kashmir, the Deccan, and eastern and western India. What needs to be emphasized is that these missionaries transmitted the message of Islam through love and without the support of the armed might of the State. The sword doubtless won converts but it also evoked hostility; the saints used persuasion and they still have a grateful and devoted following even among non-Muslims.
The socio-political conditions in India had changed by the time Ghazni invaded the country. Infighting between rival kingdoms had weakened Indian resistance. The Muslim rulers were in India not only for the spread of Islam, but had other interests also. Some of them took upon themselves the mission of Islamizing society more seriously than others; discriminatory practices such as the imposition of jizyah (poll tax), in addition to kharaj (tax on land and property), were pursued more vigorously by some rulers, while others were relaxed about them.
Despite long years of Muslim rule the overwhelming majority of the people remained Hindu. The army, state administration, and trade and commerce all depended on the direct and indirect support of the Hindus. Thus, in respect of the hated jizyah we find that it was levied sometimes and then abolished, only to be re-levied by another zealot. In fact, Islam was being Indianized; in the process, it acquired some distinctive characteristics in India. In the realms of art and architecture, philosophy and religion, medicine and other secular knowledge, there was considerable interchange. A composite culture was gradually evolving.
It may be added that Hindu rule was never completely wiped out from India. During the Delhi Sultanate, the Hindu kingdom of Vijayanagar was an impressive power in south. During Mughal times warriors like Rana Pratap refused to surrender; others worked out adjustments which left them considerable internal autonomy.
In Muslim society itself there was internal differentiation. For example, the difference between the Ashraf (those claiming descent from groups of foreign extraction) and Ajlaf (Converts from the lower Hindu castes) had a lower social position, many of the lower groups converted to Islam continued to occupy more or less their old position in society. Some Hindus occupied higher position in the royal courts and were even Generals in the army. The Hindu and Muslim aristocracy was closer. The poorer Muslims could only have the psychological satisfaction of belonging to the religion of the ruling class. But economically and socially they remained backward and exploited. The Muslim rulers of India understood the value of communal amity and realized the importance of inter community tolerance and understanding.
![]()
Question 3.
Define diversity and explain types of diversity in India.
Answer:
The term Diversity denoting collective differences so as to find out dissimilarities among the people: geographical, religious, linguistic etc. Thus all these differences presuppose collective differences or prevalence of variety of groups and culture. Indian society is characterized by unity as well as diversity.
The Nature of Diversities in India
Primarily there are major four types of diversities in India, which are;
(1) Regional Diversities: It is evident that there are extreme regional diversities in its geographic features. Indian territory contains huge mountains, thick forests, numerous rivers and etc. It is only in India that there are different regions, different types of climates and the temperatures. India is a vast country. From the Himalayas in the North to Indian Ocean in the south. There are difference in altitude, temperature, Flora and Fauna. India has every conceivable type of climate, temperature and physical configuration. There is the scorching heat of Rajastan and the biting cold of the Himalayas, Rainfall varies from 1200 to 7.5 cms per year. The result is that India has some of the wettest and driest areas in the world. India also possesses arid desserts and fertile riverine lands, bare and hilly tracts and luxuriant open plain.
(2) Linguistic Diversities: Language is another source of diversity. It contributes to collective identities and even to conflicts. The Indian Constitution has recognized 22 languages in the 8th schedule for its official purposes but as many as 1652 languages and dialects are spoken in the country. According to Grierson’s Linguistic Survey of India, these languages belong to five linguistic families, namely; Indo-Aryan languages, Dravidian languages, Austric languages, Tibeto – Burman languages and European languages.
This makes language planning and promotion difficult. But the mother tongue does evoke strong sentiments and reactions. As a consequence of this multiplicity, there is considerable bilingualism and administration has to use more than one language. Linguistic diversity has posed administrative and political challenges.
(3) Religious Diversities: There are eight major religious communities in India. Hindus constitute 82.7%, Muslims 11.8%, Christians 2.6%, Sikhs 2%,-Buddhists 0.7%, Jains 0. 4%, Zoroastrians 0.3%, and Jews 0.1%. Each major religion is sub – divided along the lines of religious documents, sects, and cults. The Hindus are now broadly divided into Shaivite (worshippers of Shiva), Vaishnavaite (worshippers of Vishnu and his incarnations), Shakta (worshippers of the Mother Goddess in various manifestations. Even among them there are sub – divisions based on doctrinal and ritual differences.
Buddhism was spread widely in India once, it lost its hold in the country of its birth and remained confined only to a few pockets. Jainism too, once held wide sway in India, and though its followers are now numerically small, they are found in both the northern and southern States. They have two main divisions: Digamber- unclothed, and Shwetamber. The Indian Muslims are divided broadly n o the Sunni and Shia communities.
Indian Christians are divided into Roman a’.holics and Protestants and into many denominational churches.
Sikhism is synthesizing religion that emphasizes egalitarianism. Grantha Sahib is the holy book and Amritsar Golden Temple is one of their holy piligrimage. The Parsis are a small community, but they have played an important role in India’s industrial development. The Jewish has been established in India for over a millennium. They also had White and Black divisions and prohibition on inter-marriage and inter-dining, but they all worshipped in the same synagogues. The Jati-like restrictions are much less in Bombay and Cochin, to where many Jews have migrated.
(4) Cultural and Ethnic Diversities: Another important source of diversity is the cultural diversity. The people differ considerably in their social habits. Cultural difference varies from state to state. The conflicting and varying shades of blood, strains, culture and modes of life, the character, conduct, beliefs, morals, food, dress, manners, social norms, Socio-Religious customs, rituals and etc. causes cultural and ethnic diversities in the country. Dr. R.K. Mukherji rightly said that “India is a museum of cults and customs, creeds and culture, faiths and tongues, racial types and social systems”. Another important source of diversity is the cultural identity of particular communities and region.
![]()
Question 4.
Define Unity and explain the factors of unity in India.
Answer:
Unity implies one-ness ora sense ofwe-ness. Meaning of integration wherein hitherto divisive people and culture are synthesized into a united whole, along with higher levels of co operation, mutual understanding, shared values, common identity and national consciousness. It lightly holds together the various relationships of ethnic groups or institutions in a neatly combined through the bonds of planned structure, norms and values.
In India aspects of Diversity & Unity co-exist, which follows as :
(1) Regional Unity: The Natural boundaries provide India a. geographical unity. In ancient times India was known as Bharatavarsha, Bharathakanda, Jambudweepa. This symbolizes the significance of historical unity. The very name “Bharatavarsha” has occupied an important place in the minds of poets, political philosophers, and religious thinkers. Each of them has conceived of the country as a single expanse from the Himalayas to Kanyakumari, a country ruled by one king Bharatha. The concept of Mother India also indicates the realization of geographical unity.
(2) Linguistic Unity: Despite the presence of number of languages, India also possesses lingual unity. Sanskrit as a common base of Indian languages provides the basis of unity as a result of which the linguistic multiplicity has been solved. Simultaneously Sanskrit became the language of Hindu culture and all classics were composed in this language, which demanded reverence and respect. People may speak different languages in different regions but they have common language of English and Hindi to communicate with each other. The formation of linguistic states and using regional languages as medium of teaching at schools, colleges and universities are the products of Independence.
In 2004 the govt, of India declared that languages that met certain requirements could be accorded the status of a classical language in India. Tamil (2004), Sanskrit (2005), Kannada (2008), Telugu (2008), Malayalam (2013) and Oriya (2014) are declared as classical languages of India. Thus it is an effort to restore linguistic heritage of India.
(3) Religious Unity: In spite of the religious diversities, it possesses religious unity. The feelings of each religious groups are the same, each accepts the truth of immortality of soul, temporary nature of world, belief in rebirth, the doctrine of karma, Salvation, Contemplation etc., There may be differences in the way these elements are treated but each religion preaches a fundamentally single religious faith and shares a belief in purity and values of life in respect of belief in unseen power, benevolence, piety, honesty and liberality, with every religious faith.
The worshippers may visit different centres of pilgrimage, but all have a common goal of “Earning religious merit by visiting a sacred place”. India is the sacred land not only for the Hindus but also for Sikhs, Jains and Buddhists. The Muslims and Christians too have several sacred centres of pilgrimage in India.
(4) Cultural Unity: In art and architecture, dress and food, literature, music and dance, sports and cinema, medicine and technology there was a fusion of style and the emergence of new forms which were the result of their combined efforts. Thus it became apparently clear from the above account that running through various diversities. India has been helped both by nature and nurture, by her geographical condition and historical experiences, by her religious ethics, and political ideas. To realize a unity to perceive, preserve and strengthen the thread of basic unity which makes India a fine example of unity in diversity, transcending birth, caste, language, ethnicity and religious groupings to establish a big society and a big nation.
Modern education, the development of a network of transport and communications, industrialization and urbanization provided new bases for unity.
![]()
Question 5.
Define National Integration and explain challenges to National Integration.
Answer:
National integration refers to national unity and a sense of belonging to the nation. It is an essential aspect in the making of a nation. Promotion of national integration is regarded as a part and parcel of the policy of any country. Many scholars have defined national integration in different ways. Among these Benjamin’s definition on national integration is quoted here According to Benjamin “National integration refers to the assimilation of the entire people of a country to a common identity”.
In simple words, National Integration refers to the process wherein a feeling of togetherness, a sense of national unity and above all, a sense of national belongingness is developed among people. It is in this context, the concept of ‘national integration’ has assumed importance. There are many challenges to national integration.
They are as follows:
(1) Regionalism: Regionalism is expressed in the desire of people of one region to promote their own regional interest at the expense of the interests of other regions. It has often led to separatism and instigated separatist activities and violent movements. It has also gained tremendous momentum during the recent years because it is exploited by the selfish politiciAnswer: Thus, regionalism has challenged the primacy of the nationalistic interests and undermines national unity. Regionalism assumed mainly four forms.
(2) Communalism: Communalism is the antagonism practiced by the members of one community against the people of community and religion. Bipin Chandra holds that communalism is the product of a particular society, economy and polity, which creates problems. Asghar Ali Engineer, Moin Shakir and Abdul Ahmed try to explain communalism as an ideological tool for propagation of economic and political interests. According to them, it is an instrument in the hands of the upper class to concentrate power by dividing people. The elites strive to maintain a status quo against transformation by dividing people on communal and religious lines.
(3) Linguism: Linguism implies one-sided love and admiration towards one’s language and a prejudice and hatred towards others’ languages. India is a land of many languages and it has been called as a ‘museum of languages’. Diversity of languages has also led to linguism. It has often been manifested into violent movements posing threat to national integration. Linguistic tensions are prevailing in the border areas which are bilingual.
(4) Extremism and Terrorism: Extremism and terrorism have emerged during the recent years as the most formidable challenges to national integration. Extremism refers to the readiness on the part of an individual or group to go to any extreme even to resort to undemocratic, violent and harmful means to fulfil one’s objectives. In the past India has been facing the problems of terrorism since independence. India has faced this problem in Nagaland (1951), Mizoram (1966), Manipur (1976), Tripura (1980) and West Bengal in (1986).
Terrorism in India is essentially the creation of politics. According to According to Prof. Rama Ahuja there are four types of terrorism India, (1) Khalistan oriented terrorism in Puniab (2) Militants terrorism in Kashmir. (3) Naxalite terrorism in west Bengal, Bihar, Madhya Pradesh, Orissa, Andhra Pradesh Telangana, Maharastra, Uttarapradesh Jharkhand, chattisghad out of 318 district 77 districts are highly Naxal poore districts causing lot of blood shed in these areas. (4) ULFA terrorism in Assam.
The Khalistan oriented Sikh terrorism was based on a dream of theocratic state, Kashmir militants are based on their separate identity. The Naxalite terrorism is based on class enmity. Terrorism in North Eastern India is based on the identity crisis and the grievance situation. In addition to these factors, corruption, poverty, unemployment/youth unrest, widening gap between rich and poor, which are also the major challenges for national integration.
![]()
Question 6.
Explain national population policy
Answer:
In 1977, ‘family planning’ was renamed as “family welfare”. The Government of India adopted the UNPF (United Nations Population Fund) guideline of delaying the -first child and spacing the subsequent birth(s);
The Primary health Centres are engaged in family planning programmes, perform two specific functions: providing services to the people and disseminating information about these services in an effective manner in order to motivate the people to accept family planning. The major objective of family planning is “To increase individual happiness and to enhance health of the society”.
National Population Policy 2000 [NPP-2000]: is the latest in the series. It reaffirms the commitment of the government towards administering family planning services. The object of NPP-2000 is to bring the total fertility rate (TFR) to replacement levels by 2010. It contains the goals and the target to be achieved by 2010.
They can be briefed here.
![]()
Question 7.
Define Demography. Explain the characteristics of demographic profile of India.
Answer:
Demography is the systematic study of population. The term Demography is derived from two Greek words i.e. demos (people) and graphein (describe), implying the description of people. The term Demography was coined by Achille Guillard in 1855. Demography studies the trends and processes associated with population including – changes in population size; patterns of births, deaths, and migration; and the structure and composition of the population, such as the relative proportions of women, men and different age groups.
There are different varieties of demography, including Formal demography which is a largely quantitative field, and Social demography which focuses on the social, economic or political aspects of population. All demographic studies are based on processes of counting or enumeration – such as the census or the survey – which involve the systematic collection of data on the people residing within a specified territory.
The American census of 1790 was probably the first modem census, and the practice was soon taken up in Europe as well in the early 1800s. In India, census was conducted by the British Indian government between 1867-72, and regular ten yearly (decennial) censuses have been conducted since 1881. Independent India continued the practice, and seven decennial censuses have been conducted since 1951, the most recent being in 201L Demographic data are important for the planning and implementation of state policies, especially those for economic development and general public welfare.
The Major Characteristics of the Demographic Profile of India:
(1) Size and Growth of India’s Population: India is the second most populous country in the world after China. According to 2011 census India’s population is 121 crores (1.21 billion). Between 1901-1951 the average annual growth rate did not exceed 1.33%, a modest rate of growth. In fact between 1911 and 1921 there was a negative rate of growth of-0.03%. This was because of the influenza epidemic during 1918-19 : 5% of the total population of the country. The growth rate of population substantially increased after independence from British rule going up to 2.2% during 1961-1981. Since then although the annual growth rate has decreased it remains one of the highest in the developing world.
(2) Age Structure of the Indian Population: India has a very young population – that is, the majority of Indians tend to be young, compare to most other countries, the share of the less than 15 age group in the total population has come down from its highest level of 42% in 1971 to 29% in 2011. The share of the 15-60 age group has increased slightly from 53% to 63%, while the share of the 60+ age group is very small but it has begun to increase (from 5% to 8%) over the same period. But the age composition of the Indian population is expected to change significantly in the next two decades. 0-14 age group will reduce its share by about 11% (from 34% in 2001 to 23% in 2026) while the 60 plus age group will increase its share by about 5% (from 8% in 2001 to about 12% in 2026).
(3) The Declining Sex-Ratio in India: The sex ratio is an important indicator of gender balance in the population. The sex ratio is defined as the number of females per 1000 males. The trends of the last four decades have been particularly worrying – from 941 in 1961 the sex ratio had fallen to an all time low of 927 in 1991 before posting a modest increase in 2001.
According to the Census of India 2011 sex ratio has been increased and now it is 940 females per 1000 males. But what has really alarmed demographers, policy makers, social activists and concerned citizens is the drastic fall in the child sex ratio. The sex ratio for the 0 – 6 years age group (known as the juvenile or child sex ratio) has generally been substantially higher than the overall sex ratio for all age groups, but it has been falling very sharply. In fact the decade 1991-2001 represents an anomaly in that the overall sex ratio has posted its highest ever increase of 6 points from the all time low of 927 to 933, but the child sex ratio in 2011 census has dropped from 927 to 914, a plunge of 13 points taking it below the overall sex ratio for the first time.
(4) Increasing Literacy Rate of Indian Population: Literacy varies considerably across gender, regions, and social groups. As can be seen from Table No. 4, the literacy rate for women is almost 22% less than the literacy rate for men. However, female literacy has been rising faster than male literacy, partly because it started from relatively low levels. Female literacy rose by about 11.2 percent between 2001 and 2011 compared to the rise in male literacy of 6.2 percent in the same period.
Female literacy was 8.9% in 1951 has increased to 65.4 in 2011 male literacy in the same period wan 27.2% has increased to 82.17. In 1951 total literacy rates 18.3% has increased to 74.04 in 2011.
(5) Increasing Rural-Urban Differences: According to 2011 Census, 68.8% population lives in rural areas while 31.2% people live in urban areas. The urban population has been increasing its share steadily, from about 17.3% in 1951 to 31.2 in 2011, an increase of about two-and-a-half times. The results of the 2001 census fully validate the diagnosis of India’s population problem in terms of the dominance of BIMARU States (Bihar, MP, Rajastan, UP) accounted for 39% of India’s population, 42% of growth and 48% of the total Illiterate population and the adverse role of these BIMARU states.
To take note of the alarming data from 2001 census to coin another acronym – DEMARU – where D stands for daughter and MARU stands for killing. In English “E” will denote elimination. Punjab, Hariyana, Himachal pradesh & Gujarath, Maharashtra as DEMARU states, where the sharp decline in the Juvenile sex ratio. These states are in the perverse practice of Foeticide. The unholy alliance between Tradition (Son complex) and Technology (Ultra sound etc.) is playing havoc with Indian society pre-birth sex determination tests & sex selective abortion are rampant in these states. During the last decade in Punjab, the sex ratio (0-6 age group) declined from 875 to 793.
In Hariyana the sex ratio (0-6 age group) decreased from 879 to 820. In Maharashtra every single district showed a decline in the Juvenile sex ratio between 1991-2001. In Himachal pradesh from 951 to 845. In Gujarath from 928 to 878. In Chandigarh from 899 to 845 and in Delhi from 915 to 865. Even though an increase in the sex ratio for the total population was 927 in 1991 to 933 to 2001. In contrast the sex ratio of child population (0¬6 age group) which was 945 in 1991 decreased to 927 in 2001. In short the girl child (below 6 years) has lost out badly.
The state-level child sex ratios offer even greater cause for worry. As many as six states and union territories have a child sex ratio of under 900 females per 1000 males. Punjab is the worst off with an incredibly low child sex ratio of 793 (the only state below 800), followed by Haryana, Chandigarh, Delhi, Gujarat and Himachal Pradesh. Uttaranchal, Rajasthan, Uttar Pradesh and Maharashtra are all under 925, while Madhya Pradesh, Goa, Jammu and Kashmir, Bihar, Tamil Nadu, Karnataka and Orissa are above the national average of 927 but below the 950 mark. Even Kerala, the state with the best overall sex ratio does not do too well at 963, while the highest child sex ratio of986 is found in Sikkim.
![]()
Question 8.
Explain the Demographic profile of Karnataka.
Answer:
According to 2001 census, Karnataka with an area of 1,91,791 sq. km. has a population of 52,850,562 with 26,898,918 males and 25,951,644 females. According to 2011 Census, the Population of Karnataka has increased to 6,10,95,297 (Males – 3,09,66,657; Females – 3,01,28,640)withasexratioof973 females for every 1000 males. Karnataka with a population of 6,10,95,297, retains the ninth rank as in 2001, in pupulation among all the 28 States, and seven Union Territories (including the National Capital Territory of Delhi) and accounts for 5.05 per cent of Country’s pupulation of 1,21,05,69,573 in 2011.
1. Rural – Urban population in Karnataka: Among the districts within the State, Bengaluru District is the most pupulated District with 96,21,551 persons and accounts for 15.75 percent of the State’s total pupulation while Kodagu District with a pupulation share of 0.91 per cent is the least pupulated District.
In terms of percentage, 61.33 per cent are Rural residents and 38.67 per cent are Urban residents. In terms of urbanization, the State has witnessed an increase of 4.68 per cent in the proportion of Urban pupulation in the last decade. Among the districts, Bengaluru is the most urbanized District with 90.94 per cent of its pupulation residing in Urban areas followed by Dharwad District (56.82 per cent), Dakshina Kannada District (47.67 per cent), Mysuru District (41.50 per cent) and Ballari District (37.52 per cent). The least urbanized District in the State is Kodagu with 14.61 per cent, preceded by Koppal District (16.81 per cent), Mandya District (17.08 per cent), Chamarajanagar District (17.14 per cent) and Yadgiri District (18.79 per cent).
During the decade 2001-11, the State population witnessed a net addition of 82,44,735 persons to its 2001 population of 5,28,50,562. Among the districts, Bengaluru District, has witnessed the highest decennial growth rate of 47.18 per cent followed by Yadgir, the newly created District, with 22.81 per cent.
Chikkamagaluru District, a predominantly plantation area in the Malnad region, is the only District in the State which has registered a negative growth rate of -0.26 per cent. Kodagu District another plantation area in the Malnad region with a growth rate of 1.09 per cent ranks 29, just above Chikkamagaluru District.
2. Sex Rartio in Karnataka: The Sex Ratio in Karnataka has increased from 965 in 2001 to 973 in 2011. The Sex Ratio for Scheduled Caste and Scheduled Tribe population is identical at 990 and is significantly higher than that of the State. Among the districts, the highest overall Sex Ratio of 1094 is recorded in Udupi District and the lowest of 916 is recorded in Bangalore District. Female population is higher than male population in Chikmagalur, Kodagu (1019), Hassan, (1012) Dakshina Kannada (1020) and Udupi (1094).
Inspite of favourable Sex Ratio, it has declined in Udupi (-36) and Dakshina Kannada (2).
3. Density in Karnataka: Density of Population: The number of persons’for every square Km.area is called the density of population. According to 2001 census Bengaluru Urban District has registered the highest density of 2,985 persons per sq. km and the lowest density per sq.km, was recorded in Kodagu (134) and Uttara Kannada (132) districts. The density of population of the state was 319 in 2011 as against 276 in 2001. The density of population of Bengaluru metropolitan city was 4,378 in 2011 as against to 2985 in 2001. Uttar Kannada (140) and Kodagu (135) have the lowest density of population in the State.
4. Seheduled caste population in Karnataka: The Scheduled Caste population in the State has increased from 85,63,930 in 2001 to 1,04,74,992, in 2011, registering a decennial growth rate of 22.32 per cent. The Scheduled Caste population constitutes 17.15 per cent of the total population of the State. The highest proportion of Scheduled Caste population is returned from Kolar District with 30.32 percent, followed by Chamarajanagar District with 25.42 per cent. The least proportion of Scheduled Caste population is recorded in the coastal district of Udupi (6.41 per cent) District.
5. Seheduled Tribe population in Karnataka: The Scheduled Tribe population in the state has increased from 34,63,986 in 2001 Census to 42,48,987 in 2011, registering a decennial growth rate of22.66 per cent. The proportion of the Scheduled Tribe population to total population of the State is 6.95 per cent. The highest proportion of Scheduled Tribe population is in Raichur District (19.03 per cent) and the least proportion is returned from Mandya District (1.24 per cent).
6. Literacy Rate in Karnataka: Literacy Rate of the State has increased from 66.64 per cent in 2001 to 75.36 per cent 2011. While the Male Literacy has increased from 76.10 per cent to 82.47 per cent, the Female Literacy rate has increased from 56.87 per cent to 68.08 per cent.
Among the districts, Dakshina Kannada District with overall Literacy rate of 88.57 per cent retains its top position, closely followed by Bengaluru District (87.67 per cent) and Udupi District (86.24 per cent). The lowest overall Literacy rate of 51.83 per cent is recorded in the newly created Yadgir District, preceded by Raichur District which has recorded 59.56 per cent.
![]()
You can Download Chapter 4 Constitution and Government Questions and Answers, Notes, 1st PUC Political Science Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
Question 1.
Write the root word of constitution?
Answer:
The root of the term constitution is the latin word ‘Constituere’.
Question 2.
What is constitution?
Answer:
The rules and principles according to which the government of a country functions is called constitution.
Question 3.
What is constitutional government?
Answer:
According to K.C. Where “Constitution is collection of legal rules which govern the government of the countries and which have been embedded in a document”.
Question 4.
What is written constitution?
Answer:
A constitution which is written down in one or several documents is called written constitution.
![]()
Question 5.
What is unwritten constitution?
Answer:
A constitution which is not written down but has grown during various centuries is called unwritten constitution.
Question 6.
What is rigid constitution?
Answer:
A constitution which cannot be amended easily is called rigid constitution.
Question 7.
What is flexible constitution?
Answer:
A constitution which can be amended easily is called flexible constitution.
Question 8.
Give the best example for written constitution.
Answer:
America, India having written constitution.
Question 9.
Give the best example for un-written constitution.
Answer:
England having un-written constitution.
Question 10.
Give the best example for rigid constitution.
Answer:
America is having rigid constitution.
Question 11.
Give the best example for flexible constitutions.
Answer:
England is having flexible constitution.
Question 12.
What is democratic government?
Answer:
A government which can be ruled by the people is called a democratic government.
Question 13.
Give the meaning of direct democracy.
Answer:
A government where people are directly participating in the administration is called direct democracy.
Question 14.
Give an example of direct democracy.
Answer:
Ancient Greek city-states having direct democracy.
![]()
Question 15.
Give the meaning of indirect democracy.
Answer:
A government where the representatives are participating in the administration is called indirect democracy.
Question 16.
Give an example for indirect democracy.
Answer:
India is having indirect democracy.
Question 17.
What is dictatorial government?
Answer:
A government where only one person or group run the administration is called a dictatorial government.
Question 18.
Give an example for dictatorial government.
Answer:
Germany was an example for the dictatorial government.
![]()
Question 19.
What is ancient dictatorship?
Answer:
To meet the immediate crisis dictators came into existence is called ancient dictatorship.
Question 20.
Give an example for ancient dictatorship.
Answer:
Ancient Rome was a example of ancient dictatorship.
Question 21.
What is modern dictatorship?
Answer:
Once a dictator came to power is desiring to be in the power permanently is called modem dictatorship.
Question 22.
Give an example of modern dictatorship.
Answer:
Saddam Hussian of Iraq was an example for modern dictatorship.
Question 23.
What is the root word of democracy?
Answer:
The word democracy has been derived from the Greek words ‘Demos’ and ‘Kratia’.
Question 24.
Define parliamentary government.
Answer:
In parliamentary form of government executive is the part of legislature and responsible to the legislature.
Question 25.
Give an example for the parliamentary government.
Answer:
England is an example of parliamentary government.
![]()
Question 26.
What is presidential government?
Answer:
In this system, the chief executive is directly elected by the people. It is not the part of the legislature.
Question 27.
Give an example for the presidential government.
Answer:
America is an example for the presidential government.
Question 28.
What is the root word of the federation?
Answer:
The root word of federation is the Latin word ‘foedus’.
Question 29.
What is the federal government?
Answer:
A government where the powers have been distributed between union and state is called federal government.
Question 30.
Give an example for federal government.
Answer:
America is an example for federal government.
Question 31.
What is unitary government?
Answer:
A government where the powers are concentrated in union is called unitary government.
Question 32.
Give an example for unitary government.
Answer:
England is an example for unitary government.
Question 33.
Who used the word constitution for the first time?
Answer:
The word constitution was used by Henry II of England.
Question 34.
What is constitutional law?
Answer:
Constitutional law is a fundamental law of a country drafted and enacted by constitutional assembly.
Question 35.
What is the ordinary law?
Answer:
The ordinary law is not fundamental law. It is subordinate to the constitutional law.
![]()
Question 36.
What is constitutional government?
Answer:
Government which functions on the basis of the law laid down by constitution is constitutional government.
Question 37.
What is ideal constitution?
Answer:
Burgess describes an ideal constitution as one which contains the principle of liberty, government and sovereignty.
Question 38.
What is meant by collective responsibility?
Answer:
It means the cabinet and council of ministers individually and collectively responsible to the parliament:”
Question 39.
What is “no-confidence motion”?
Answer:
It is the power of the legislature to remove the ministry from office.
Question 40.
What is veto-power?
Answer:
It is the power of President to say ‘No’ to a law passed by parliament’ and send it back for reconsideration.
Question 41.
Which is the lengthiest constitution in the world?
Answer:
Indian constitution.
Question 42.
Who is the protector of constitution?
Answer:
The Judiciary (Supreme court).
Question 1.
Define constitution.
Answer:
According to K.C. Wheare “Constitution is collection of legal rules which govern the government of the countries and which have been embedded in a document”.
Question 2.
Write the meaning of constitutional government.
Answer:
A government which functions in accordance with defined rule as laid down in constitution is known as constitutional government. In short constitutional government means limited government.
Question 3.
Define written constitution.
Answer:
According to C.F. Strong “A written constitution is in the form of a document which has a special sanctity”.
Question 4.
Define un-written constitution.
Answer:
According to Sir Ivor Jennings – “An unwritten constitution consists of institutions and not paper documents. It is not made but has grow n from time to time by a process of evolution”.
Question 5.
Define rigid constitution.
Answer:
According to A.V. Dicey a rigid constitution is one which certain laws generally are known as constitutional or fundamental laws cannot be changed in the same maimer as the ordinary laws.
Question 6.
Define flexible constitution.
Answer:
According to Gamer the flexible constitution places constitutional law and ordinary law on the same footing.
![]()
Question 7.
Write the meaning of democratic government.
Answer:
Democracy is the form of government in which the ruling power is largely vested not in any particular class or classes but in the members of the community as a whole.
Question 8.
What do you mean by the dictatorial government?
Ans.
A form of government in which absolute power is concentrated in a person or group is called dictatorial government.
Question 9.
Define parliamentary government.
Answer:
In parliamentary form of government executive is the part of legislature and responsible to the legislature.
Question 10.
What is the presidential government?
Answer:
In this system, the chief executive is directly elected by the people. It is not the part of the legislature.
Question 11.
Define federal government and give example.
Answer:
It is the government which the powers of the state are divided and distributed between central and state government. It is, also known as dual polity. The federal government discharges its power and authority in accordance with constitution. Eg: U.S.A
Question 12.
What is meant by unitary system of government?
Answer:
In the unitary system of government, where all the powers and authority of the state is concentrated in the single Central government and the supreme power is exercised by it.
Eg: England, France, Japan, etc.
Question 13.
Define and explain the term constitution.
Answer:
According to Lord Bryce “The constitution is the aggregate of laws and customs under which the life of states goes on” Maclver defines as “law which governs the state”.
Question 14.
What are the characteristics of democratic government?
Answer:
Question 15.
What is presidential form of government?
Answer:
In this system, the legislature and executive are separate and independent. The term of office of the president is fixed and goes by calender.
![]()
Question 16.
How do non-democratic government arise?
Answer:
It arises out of illegal method. It is the result of the use of force. A powerful individual or group can overthrow the constitutional government and establish dictatorship. ,
Question 17.
Describe the features of the Presidential system of government.
Answer:
In Presidential executive president is the supreme head of the state. Here executive is neither elected, nor responsible to legislature. The term of office here is fixed. It is not responsible to the legislature.
Question 18.
Describe the merits of Parliamentary form of government.
Answer:
Question 19.
What are the demerits of parliamentary form of government?
Answer:
Question 20.
Distinguish between division of powers and separation of powers.
Answer:
Division power is in a federal form of government Power is divided between the central and state government, where as separation of powers means there is clear cut demarcation between 3 organs of government that is Legislature, executive and judiciary.
Question 21.
Give one definition of democracy.
Answer:
According to Lord Bryce “Democracy is the form of government where the ruling power of the state is legally vested not in any particular class but in members of community as a whole.
Question 22.
What is the meaning of federal government?
Answer:
The word federal is derived from the Latin word ‘Foedus’, which means treaty or agreement. The federal form of government comes into existence through treaty or agreement, when a state with its vast geographical territory, heterogeneous characteristics desires to protect them is born by creating new state.
Question 23.
What is Indirect democracy? Give example.
Answer:
It is also known as representative democracy. The citizens of a state elect their representatives in the election. The elected representatives administer the country on behalf of people. Eg : India, U.S.A, U.K.
Question 24.
What is the difference parliamentary and presidential form of government?
Answer:
The distinction between parliamentary and presidential executive is based on the relationship between the legislature and executive. If the executive depends on legislature for its survival it is a parliamentary government and if the executive is independent of legislative it is presidential executive.
Question 1
Explain the significance of constitution.
Answer:
The significance of a constitution to the state is discussed below:
1. Outlines the goals of state:
No state can progress and prosper, however rich and wealthy, if it does not possess clear cut aims and objectives. A constitution details the path to be followed by a state in accomplishing its objectives thereby living up to the aims and objectives of its people.
2. Regulates behavior of people:
The constitution provides for basic guidelines or a reasonable code of conduct which helps create a civil and civilized society. A constitution is very important in harmonizing relationship between and among numerous sections of people.
3. Control over authority of the government:
The government derives its power from the constitution and is bound to abide by provisions of the constitution. No government can take law into its hand and deal whimsically.
4. Establishes ruler – ruled relationship:
The people and government are like two faces of the same coin. A state can succeed only if people and government share mutual trust and co-operation. The constitution should avoid any kind of needless friction between the governor and the governed.
5. A guide to future generations:
A constitution is often referred to as the mirror of a state which reflects the basic ambitions and aspirations of a state. By acting as a standard of reference it guides future generations to make necessary changes based on the existing structure.
Question 2.
Describe the Characteristics of constitutional government.
Answer:
The Characteristics of constitutional government are as below:
1. Supremacy of the constitution :
In this system all the powers of government are laid down in accordance with the provisions of constitution. Government should function with in the frame work of the constitution.
2. Equality before law :
Here law will apply to all the people equally. The benefits of law can be enjoyed all the citizens irrespective of caste, creed, religion, social and economic
status.
3. Individual liberty :
In the constitutional government all the citizens are enjoying the fundamental rights guaranteed by the constitutions. So constitution provides the rights to all the people and ensures protection.
4. Protects public interest :
All people in the country should be lived under the purview of constitution. Violation of the provisions of constitution invites severe punishment. So constitution safeguards the interests of the people by protecting them from all the outside aggression.
Question 3.
Explain the features of wirtten constitution.
Answer:
The features of written constitution are explained below.
Question 4.
Explain the features of unwritten constitution.
Answer:
The features of unwritten constitution are explained as below.
Question 5.
Explain the features of rigid constitution.
Answer:
The features of rigid constitution are explained below.
Question 6.
Explain the features of flexible constitution.
Answer:
The features of the flexible constitution are explained as below
Question 7.
Describe the essential elements of the Ideal constitution.
Answer:
The essentials of an ideal constitution are explained as below
1. It should be definite :
An ideal constitution should not be vague but clearly narrate the provisions which relates to the organization of the government. The principles should be precise and clarity.
2. It should be comprehensive:
An ideal constitution must be comprehensive enough to mention the functions of the government and rights, duties of the citizens. The constitution should not be too big but include all the information on the government.
3. Method of amendment:
An ideal constitution should possess the method of amendment. As the social condition of the people is going on change, the constitution must also undergoes change. It should represent the future needs of the future generation.
4. It should correspond to reality :
An ideal constitution should correspond to the real, conditions obtained with in the state, otherwise it cannot work properly.
![]()
Question 8.
Explain the features of unitary governments.
Answer:
Features of Unitary Government:
1. Concentration of Power:
A Unitary government is characterized by the presence of a single centre, which is omnipotent and omnipresent all over the territory. All decisions of the state flow from one single centre.
2. No Provincial Autonomy:
The provinces or local units in a unitary system are created by the centre for the sake of administrative convenience. It carries out the orders of the centre without having any powers to make decisions. Thus, the local units only act as subordinate agents of the centre without any authority or autonomy.
3. Single legislature:
In a Unitary system of government there will be only one single supreme legislative assembly which makes laws for the whole country and are faithfully implemented by the local units.
4. Constitution may be written or unwritten:
The constitution, in a unitary government, may be written or unwritten as there is one single central authority wielding power all over the state without any other centres of power.
Question 9.
Explain the features of federal government.
Answer:
1. Division of Powers:
A federal government is characterized by the existence of two governments- the centre and the local government created on the basis of division of powers. Both are independent and autonomous within their spheres of powers and yet interdependent. The residuary powers rests with the centre in some states (e.g. India)
and in the hands of local units in some states (e.g. Great Britain).
2. Supremacy of the constitution:
In a Federal system, the power enjoyed by the centre and local units is original because both derive their powers from the constitution. This avoids any confusion or contention in the sharing of power. The division of powers is based on necessity and convenience.
3. Written and rigid constitution:
The constitution in a federal government would definitely be rigid because it has to deal with powers related to both center and local governments. Each and every detail should be explained in a written form in order to avoid any clash of jurisdiction or possible over-lapping of responsibilities. The amendment procedure would be rigid which protects the interest of the federation from frequent, mindless changes to the constitution.
4. Special provision for settlement of disputes:
In a presidential system, in order to settle disputes arising between the states or between centre and the states, the judiciary has been assigned the job of interpreting the provisions of the constitution, thus acting as custodian and guardian of the constitution.
5. Power of amendment:
In a presidential system, to amend the constitution both the centre and the local governments have been assigned equal powers. No constitutional amendment can be made without the consent of federal units.
Question 10.
Distinguish between federation and confederation.
Answer:
Confederation is an association of state founded on the basis of common interests. The member states, retain their original sovereignty and independence.
Federation is permanents established through a constitution. Constitution units do not possess the right to withdraw from a federation.
Question 11.
What is residuary powers?
Answer:
In a federal government subjects of national interest are allocated to the centre. Matters of regional or local interests are given to units or states. The remaining powers those which have not been mentioned in the constitution are known as residuary powers.
![]()
Question 12.
Describe the parliamentary type of executive.
Answer:
There is a difference between real and nominal executive head on whom all powers are concentrated, but these powers are exercised by the cabinet. In this system, cabinet is responsible to the parliament for all their actions. There is individual and collective responsibility of ministers. There is a close relationship between the cabinet and the council of ministers.
Question 13.
Explain the characteristic of Constitution.
Answer:
Question 14.
What is a Constitutional Government?
Answer:
A government that is run strictly in accordance with provisions of the constitution is called a constitutional government. Constitutional governments It strives for the welfare of the whole society rather than any perceived vested interest. These governments are a creation of the people and therefore obedience and allegiance to it are natural. The use of force in running a government does not find a place in a constitutional government. Constitutional government is also called “limited government” or “controlled government”.
Question 15.
What is Democracy? Explain the features of Democracy?
Answer:
The word “Democracy” is a derivative of Greek words ‘Demos’ and ‘Kratia’ meaning ‘people’ and ‘rule’ respectively. Thus, democracy is the rule of the people.
Features of Democratic Government:
The following features can be identified by democratic form of government.
Question 16.
What is Dictatorship? What are the kinds of dictatorship?
Answer:
Dictatorship is a form of government where all the powers of the state are concentrated in one man or in one party or in one group. Dictatorship is classified as traditional and modem, old and new, leftisit fascist, communist and military. The old type of dictatorship existed in ancient Greece and Rome; they are temporary and appointed during crisis. Modem dictatorship is of new type and permanent. Modem dictatorship has taken three forms;
Question 17.
What is a Federal Government?
Answer:
In a federal government, there will be two sets of government. The powers and authority of government is divided between a government for the whole country and provincial governments of the country in such a way that each government is legally independent within its own sphere.
The term ‘Federation’ is derived from the Latin word ‘Foedus’ meaning ‘treaty’ or ‘agreement’. Two or more provinces, after surrendering the sovereignty, on agreement agree to work within the union with a certain degree of autonomy under a single sovereignty. The Central government and the provincial governments are neither superior nor subordinate to each other but work in unison with co-ordination and co-operation.
The best examples for a federal form of government are the USA, India, Switzerland, Australia and Canada.
Question 18.
Explain Division of powers in India.
Answer:
In India, the division of powers is based on three lists i.e., the Union list, State list and Concurrent list. The Union list consists of 99 Subjects such as defense, national security; home affairs, science and technology are vested in the hands of central government. The State List consisting of 66 subjects like education, health, maintenance of law and order, providing basic facilities etc., are vested in the state governments. There are certain provisions in which both states and centre shall share power, under the Concurrent list consisting of 52 subjects such as marriage, divorce, contracts etc.
Question 1.
Write the meaning and significance of constitution.
Answer:
Importance of a constitution to the state is discussed below:
1. Outlines the goals of state:
No state can progress and prosper, however rich and wealthy if it does not possess clear cut aims and objectives. A constitution details the path to be followed by a state in accomplishing its objectives thereby living up to the aims and objectives of its people.
2. Regulates behavior of people:
The constitution provides for basic guidelines or a reasonable code of conduct which helps create a civil and civilized society. A constitution is very important in harmonizing the relationship between and among numerous sections of people.
3. Control over the authority of the government:
The government derives its power from the constitution and is bound to abide by provisions of the constitution. No government can take law into its hand and deal whimsically.
4. Establishes a ruler — ruled relationship:
The people and government are like two faces of the same coin. A state can succeed only if people and the government share mutual trust and co-operation. The constitution should avoid any kind of needless friction between the governor and the governed.
5. A guide to future generations:
A constitution is often referred to as the mirror of a state which reflects the basic ambitions and aspirations of a state. By acting as a standard of reference it guides future generations to make necessary changes based on the existing structure.
![]()
Question 2.
Distinguish between written and unwritten constitution.
Answer:
Written constitution
Unwritten constitution
Question 3.
Distinguish between rigid and flexible constitution.
Answer:
Rigid constitution
Flexible Constitution
Question 4.
Describe the features of democratic government.
Answer:
1. The government in a democracy is responsible to the people. The government will also have to function according to public opinion. Self-government makes the people more disciplined and there will be more responsible citizens than in any other form of government.
2. Democracy upholds the principles of liberty and equality. Political and economic equality are assured in a democracy.
3. Democracy respects the dignity of human being. It provides rights and liberties for the development of the personality of individual.
4. A democratic government promote the welfare of the people, where as in other forms of government only particular class may be benefitted.
5. Democratic government is stable and efficient government. It avoids the revolution because it tarried on according to the wishes of the people.
6. Unlike other forms of government, democracy is self-corrective. In democracy the freedom of speech and freedom of press creates an enlightened public opinion
7. Democracy is progressive and educative force. In a democracy, people have full civil and political rights. It is a training ground for active, healthy and intelligent citizenship.
8. In democracy, there is order, peace and progress. It is flexible government which adopts itself to change peacefully.
9. As the people have a share in the government of country, the spirit of patriotism is strengthened and everybody is willing to work and undergo sacrifices for the welfare of the community.
10. Democracy protects the minority. A written constitution guarantees the rights of the minorities.
Question 5.
Explain the features of the dictatorial government.
Answer:
1. Absolute power:
Dictatorship is characterized by absolute power where the dictator controls the constitution. He can make and unmake laws. All the laws must originate from him and there is neither a limit on his tenure nor is he subjected to any other authority.
2. Based on Force:
Dictatorship stands on the twin pillars of force and coercion. The word of the dictator should be honored in letter and spirit. Any violation of the order may result in severe punishment or even death.
3. Totalitarian state:
Dictatorial regimes regulate and control all aspects of human existence. It provides security, basic necessities such as food, shelter and clothing, education and order in society. In totalitarian state’s individual personality is suppressed and all aspects of an individual are regimented and brought under the control of the state. The totalitarian approach is well summarized by Mussolini when he says: “Everything within the state, nothing above the state, nothing outside the State’’. Thus, the State is the central point around which all human activities must revolve.
4. One Nation one party:
In a dictatorship, for the whole state, there is only one constitution and the administration is managed by one single party and that is responsible for the whole state. Any kind of criticism of the party or the leadership is not tolerated. The distinct feature of dictatorship is its intolerance to criticism and new ideas.
5. No individual liberty:
In a dictatorship, individual freedom and liberty do not find place. Freedom of thought and expression is restricted.
Question 6.
What is a parliamentary form of government? Explain its features.
Answer:
A parliamentary form of government is characterized by the direct responsibility of the executive to the legislature which lasts as long as it enjoys the confidence of the legislature. The parliamentary government system which first began in England (Great Britain) is also called as ‘Cabinet government’ and ‘Responsible government’.
Features of a Parliamentary form of Government.
1. Nominal Executive:
The presence of a nominal executive is a primary feature of parliamentary government. Though all powers of the state are vested in his name by the constitution, it is in fact exercised by the real executive consisting of Prime minister and council of ministers. For example, the Queen of England and the President of India are the nominal executives.
2. Collective responsibility:
A parliamentary government is directly responsible to the legislature and stays in office as long as it enjoys the confidence of the lower house. A decision taken by cabinet is binding on ministers and they should defend it in and out of parliament. Together we sink or sail” is the key statement of parliamentary government.
3. Individual Responsibility: Each minister will be in charge of a ministry and is wholly responsible for all decisions of the ministry to the legislature. Parliamentary government and individual responsibility go hand in hand.
4. Political Homogeneity:
In a parliamentary government, all ministers belong to the same party or alliance having common policies, programmes, values and beliefs and work under a common minimum programme (CMP). As a result, it is not difficult to secure co¬operation and co-ordination.
In case, a government consists of divergent policies and beliefs, frequent quarrels, squabbling, and infighting brings down the government. It defeats the very purpose of government formation.
5. Membership of parliament:
In a parliamentary government, a minister must be a member of either house of parliament. On becoming a minister he must become a member of either house within 180 days. Otherwise, he will have to step down from office. This is to ensure individual responsibility to the parliament.
6. Oath of Secrecy:
It is the responsibility as well as duty of the cabinet to protect official secrets. Cabinet decision should not become public without the authorization from parliament. Ministers are administered the ‘oath of secrecy’ to maintain secrecy of cabinet decisions. The purpose is to avoid any person or a group taking undue advantage of the leaked information.
7. Leadership of the Prime Minister:
In parliamentary government, Prime Minister is the backbone of the cabinet. He acts as the spokesperson of the government by defending it in and out of the government. He acts as the advisor to the president in summoning, proroguing and dissolving the parliament. The Prime minister exercises independence in appointing ministers, expanding the ministry and allocation of portfolios.
8. Effective opposition:
Opposition party is called “the government in waiting”. In parliamentary government, opposition parties follow the day-today administration and
bring before the people the failures of the government from time to time. During elections they seek a mandate on the basis of the failures of the government Thus, opposition acts as a watchdog of the government.
In fact, in Britain, the shadow cabinet functions just like the government but without powers.
They too have separate departments to monitor developments in respective areas.
![]()
Question 7.
What is a Presidential form of Government? Explain its features.
Answer:
In Presidential government the Executive is not responsible to the legislature. It is based on the principle of ‘Separation of powers’. It is known as non-responsible system or fixed executive system. E.g. USA.
Features of presidential government.
1. Separation of powers:
The Presidential government is based on Montesquieu concept of “separation of powers”. The concept of separation powers contend that the legislature, executive and judiciary must be independent of each other and function independently. Legislature performs the task of law-making, executive law implementation and judiciary interpretation of laws.
2. Checks and Balances:
The presidential government is based on checks and balances. Though the legislature, executive and judiciary function independently, complete separation is not only desirable but also impractical. To maintain the exercise of power balanced, controlled and widespread each organ is given a fair degree of power in one another ’s the functional area.
3. Executive is not responsible to legislature:
The president in U S.A is directly elected by the people and hence is not responsible to the legislature. The president can’t participate in the proceedings of the congress. He can neither initiate a bill nor pilots it. The President is not accountable to anyone but the constitution and the people.
4. Real Executive:
In the presidential government, the president is directly elected by the people. The president is not only the real executive but also the head of government. He is directly responsible for all happenings in the country.
5. Secretaries directly responsible to the president:
The secretaries known as presidents ‘Brain Trust’ are appointed by the president and stay in office as long as they enjoy the confidence of the president. The President may remove any secretary without assigning any reason. Secretaries are neither responsible to congress nor to the people but to the president. The President can hire or fire secretaries
6. Fixed Tenure:
The President does not depend on the congress for his survival. He, is directly elected for a period of 4 years and lasts his full term. He cannot be removed from office except on grounds of inefficiency and proven misbehavior through an impeachment motion.
Question 8.
Distinguish between democratic and dictatorial government.
Answer:
Democratic government :
Dictatorial government
Dictatorship is a form of government where all the powers of the state are concentrated in one man or in one party or in one group. Dictatorship is classified as traditional and modem, old and new, leftisit fascist, communist, and military. The old type of dictatorship existed in ancient Greece and Rome; they are temporary and appointed during crisis. Modem dictatorship is of new type and permanent. Modem dictatorship has taken three forms;
Question 9.
Distinguish between parliamentary and presidential government.
Answer:
A parliamentary form of government is characterized by the direct responsibility of the executive to the legislature which lasts as long as it enjoys the confidence of the legislature. The parliamentary government system which first began in England (Great Britain) is also called as ‘Cabinet government’ and ‘Responsible government’.
1. Nominal Executive:
The presence of a nominal executive is a primary feature of parliamentary government. Though all powers of the state are vested in his name by the constitution, it is in fact exercised by the real executive consisting of Prime minister and council of ministers. For example, the Queen of England and the President of India are the nominal executives.
2. Collective responsibility:
A parliamentary government is directly responsible to the legislature and stays in office as long as it enjoys the confidence of the lower house. A decision taken by cabinet is binding on ministers and they should defend it in and out of parliament. Together we sink or sail” is the key statement of parliamentary government.
3. Individual Responsibility:
Each minister will be in charge of a ministry and is wholly responsible for all decisions of the ministry to the legislature. Parliamentary government
and individual responsibility go hand in hand.
4. Political Homogeneity:
In a parliamentary government, all ministers belong to the same party or alliance having common policies, programmes, values and beliefs and work under a common minimum programme (CMP). As a result, it is not difficult to secure co¬operation and co-ordination.
In case, a government consists of divergent policies and beliefs, frequent quarrels, squabbling, and infighting brings down the government. It defeats the very purpose of government formation.
5. Membership of parliament:
In a parliamentary government, a minister must be a member of either house of parliament. On becoming a minister he must become a member of either house within 180 days. Otherwise, he will have to step down from office. This is to ensure individual responsibility to the parliament.
6. Oath of Secrecy:
It is the responsibility as well as the duty of the cabinet to protect official secrets. Cabinet decision should not become public without the authorization from cabinet decisions. The purpose is to avoid any person or a group taking undue advantage of the leaked information.
7. Leadership of the Prime Minister:
In parliamentary government, Prime Minister is the backbone of the cabinet. He acts as the spokesperson of the government by defending it in and out of the government. He acts as the advisor to the president in summoning, proroguing and dissolving the parliament. The Prime minister exercises independence in appointing ministers, expanding the ministry and allocation of portfolios.
8. Effective opposition:
Opposition party is called “the government in waiting”. In parliamentary government, opposition parties follow the day-today administration and bring before the people the failures of the government from time to time. During elections, they seek a mandate on the basis of the failures of the government. Thus, opposition acts as a watchdog of the government.
In fact, in Britain, the shadow cabinet functions just like the government but without powers.
They too have separate departments to monitor developments in respective areas.
Presidential Government:
In the Presidential government, the Executive is not responsible to the legislature. It is based on the principle of ‘Separation of powers’. It is known as a non-responsible system or fixed executive system. E.g. USA.
1. Separation of powers:
The Presidential government is based on Montesquieu concept of “separation of powers”. The concept of separation powers contend that the legislature, executive and judiciary must be independent of each other and function independently. Legislature performs the task of law-making, executive law implementation and judiciary interpretation of laws.
2. Checks and Balances:
The presidential government is based on checks and balances. Though the legislature, executive and judiciary function independently, complete separation is not only desirable but also impractical. To maintain the exercise of power balanced, controlled and widespread each organ is given a fair degree of power in one another’s the functional area. ,
3. Executive is not responsible to the legislature:
The president in U S A is directly elected by the people and hence is not responsible to the legislature. The president can’t participate in the proceedings of the congress. He can neither initiate a bill nor pilots it. The President is not accountable to anyone but the constitution and the people.
4. Real Executive:
In the presidential government, the president is directly elected by the people. The president is not only the real executive but also the head of government. He is directly responsible for all happenings in the country.
5. Secretaries directly responsible to the president:
The secretaries known as presidents ‘Brain Trust’ are appointed by the president and stay in office as long as they enjoy the confidence of the president. The President may remove any secretary without assigning any reason. Secretaries are neither responsible to congress nor to the people but to the president. The President can hire or fire secretaries.
6. Fixed Tenure:
The President does not depend on congress for his survival. He is directly elected for a period of 4 years and lasts his full term. He cannot be removed from office except on grounds of inefficiency and proven misbehavior through an impeachment motion.
![]()
Question 10.
Distinguish between federal and unitary government.
Answer:
Unitary government:
Federal government:
Question 11.
What are the kinds of Democracy?
Answer:
There are two kinds of Democracy
1. Direct Democracy:
Direct democracy is a kind of governance where the people directly ’’ take part in electing their government and also have a share in the exercise of real power. For example, the ancient Greek and Roman city-states had direct democracy where people directly participated to elect their representatives and take necessary measures.
In modem times, the direct democracy is still in practice in some of the cantons of Switzerland, where the voters meet in ‘ open-air parliament’. However, direct democracy . is best suited to only countries small in area and a population small enough to directly take part in the affairs.
2. Indirect Democracy:
Under this system, the people participate in the affairs of the state through their representatives. Though popular sovereignty rests in the hands of the people, it is actually exercised by their representatives and they are in turn responsible to the people. If the representatives fail to stand up to the aims and aspirations of the people, they may be changed in the next election.
Question 12.
What are the factors that condition the success of Democracy?
Answer:
1. Liberty and Equality:
Democracy can work satisfactorily only when people are allowed to enjoy a greater amount of freedom and equality. Freedom is necessary for the participation of the people in their administrative affairs. People must enjoy economic, social and political liberty and equality without any discrimination.
2. Eternal Vigilance:
Eternal vigilance is the price of democracy. Vigilance is nothing but. alertness or watchfulness of the people. Citizens must constantly watch as to how the government is doing its administrative business. The government can be kept on track if the people are constantly vigil on what is going on at the government level.
3.Spirited Leadership:
Spirited leadership is a must for the success of democracy. Leaders should be able, efficient, intelligent, active and honest. They must have good reasoning capacity, rational attitude, clean-hands and descent behavior. They must take proper steps to increase the competence of the government.
4. Literacy:
The active and intelligent participation of citizens in public affairs can be assured if all of them are adequately educated. The government has to chalk out plans to provide free and compulsory education for all.
5. Bi-party system:
For the successful working of democracy, there must be only two political parties.
6.Strong Opposition:
For the success of democracy, there must be a strong opposition. The government becomes irresponsible and careless in the absence of strong and effective opposition parties. Opposition parties should check the illegal, unpopular and unconstitutional act of the ministers and the representatives. They should try to keep the government on the right track through their rational criticism.
7. Participation:
The people must have an independent opinion in public affairs. The success of democracy depends upon the ability, character and power of common man.
8. Tolerance:
Tolerance is a noble virtue of democracy. The government must be tolerant enough to listen to the criticism made by people from time to time. People must be tolerant enough to understand the problems of the government. The minority must accept the majority views, and majority must understand the feelings of the minority.
9. Independent Judiciary:
In all democratic countries, the judiciary should play a significant role. An independent and impartial judiciary is necessary to protect the fundamental rights of the people guaranteed in the constitution. For the administration of justice, settlement of disputes and to interpret the laws and constitution, an independent judiciary is a must.
10. Decentralization:
It is the most important feature of democratic government. Power in the hands of the people is called decentralization. Rural and Urban Panchayat Raj system has been implemented on the basis of decentralization.
![]()
Question 13.
What is a Unitary Government? Explain its features.
Answer:
A unitary government is system in which the power and authority to make and unmake laws lie with the centre. It enjoys the authority of transferring power to provincial units, for the sake of administrative convenience, which can be installed, changed or removed at the discretion of the single central authority. The United Kingdom, France, Italy, Japan, China, Sri Lanka, and Iran are some of the examples of a unitary state.
Features of Unitary Government:
1. Concentration of Power:
A Unitary government is characterized by the presence of a single centre, which is omnipotent and omnipresent all over the territory. All decisions of the state flow from one single centre.
2. No Provincial Autonomy:
The provinces or local units in a unitary system are created by the centre for the sake of administrative convenience. It carries out the orders of the centre without having any powers to make decisions. Thus, the local units only act as subordinate agents of the centre without any authority or autonomy.
3. Single legislature:
In a unitary system of government, there will be only one single supreme legislative assembly which makes laws for the whole country and are faithfully implemented by the local units.
4. Constitution may be written or unwritten:
The constitution, in a unitary government, may be written or unwritten as there is one single central authority wielding power all over the state without any other centres of power.
Question 14.
Explain the features of a Federal Government.
Answer:
1. Division of Powers:
A federal government is characterized by the existence of two governments- the centre and the local government created on the basis of division of powers. Both are independent and autonomous within their spheres of powers and yet interdependent. The residuary powers rests with the centre in some states (e.g. India) and in the hands of local units in some states (e.g. Great Britain).
2. Supremacy of the constitution:
In a Federal system, the power enjoyed by the centre and local units is original because both derive their powers from the constitution. This avoids any confusion or contention in the sharing of power. The division of powers is based on necessity and convenience.
3. Written and rigid constitution:
The constitution in a federal government would definitely be rigid because it has to deal with powers related to both centre and local governments. Each and every detail should be explained in a written form in order to avoid any clash of jurisdiction or possible over-lapping of responsibilities. The amendment procedure would be rigid which protects the interest of the federation from frequent, mindless changes to the constitution.
4. Special provision for settlement of disputes:
In a presidential system, in order to settle disputes arising between the states or between centre and the states, the judiciary has been assigned the job of interpreting the provisions of the constitution, thus acting as custodian and guardian of the constitution.
5. Power of amendment:
In a presidential system, to amend the constitution both the centre and the local governments have been assigned equal powers. No constitutional amendment can be made without the consent of federal units.
You can Download नागरिक के कर्तव्य Questions and Answers Pdf, Notes, Summary Class 10 Hindi Karnataka State Board Solutions to help you to revise complete Syllabus and score more marks in your examinations.
अभ्यास
I. निम्नलिखित प्रश्नों के उत्तर लिखिए :
प्रश्न 1.
मीना मैडम ने कितने दिनों के कार्यशिबिर का आयोजन किया था ?
उत्तर:
मीना मैडम ने पंद्रह दिनों के कार्यशिबिर का आयोजन किया था।
प्रश्न 2.
संभाषण का विषय क्या था ?
उत्तर:
नागरिकों के मूल कर्तव्य यह संभाषण का विषय था।
प्रश्न 3.
अकुल ने मीना मैडम से क्या कहा ?
उत्तर:
अकुल ने मीना मैडम से कहा, एक नागरिक की है सियत से हमें अपने देश के राष्ट्र\(\square\)ध्वज, राष्ट्र\(\square\)गान र तथा। राष्टी \(\square\)य त्योहारों का आदर करना चाहिए। करना चाहिए।
प्रश्न 4.
सलमा ने क्या कहा ?
उत्तर:
सलमा ने कहा कि प्रकृति हमारी माँ है। इसलिए हमें प्राकृतिक संसाधनों का अपव्यय नहीं करना चाहिए। साथ ही पर्यावरण को स्वच्छ रखना भी हमारा कर्तव्य है।
प्रश्न 5.
अन्वर ने मीना मैडम से क्या कहा ?
उत्तर:
अन्वर ने मीना मैडम से कहा कि देशवासियों के प्रति भाईचारे का भाव रखना जरूरी है। जाति, धर्म, भाषा, प्रदेश, वर्ग पर आधारित सभी भेदभावों से दूर रहना चाहिए।
प्रश्न 6.
मीना मैडम ने छात्रों से अंत में क्या कहा ?
उत्तर:
मीना मैडम ने छत्रों से कहा कि देश की म रक्षा और उत्कर्ष के लिए सतत प्रयत्नशील रहें। अपने कर्तव्यों का पालन करें तभी देश का कल्याण होगा।
You can Download Chapter 3 Basic Political Concepts Questions and Answers, Notes, 1st PUC Political Science Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
Question 1.
What is Sovereignty?
Answer:
Sovereignty is the supreme power of the state.
Question 2.
Which is the most essential element of the state?
Answer:
Sovereignty is the most essential element of the state.
Question 3:
Which is the root word of sovereignty?
Answer:
The word sovereignty has been derived from the Latin word ‘ Superanus’.
![]()
Question 4.
What are the two kinds of Sovereignty?
Answer:
Internal and External Sovereignty.
Question 5.
Who introduced (aspects) the sovereignty in the modern sense?
Answer:
Jean Bodin introduced Sovereignty in the modem sense.
Question 6.
Name the book written by Jean Bodin.
Answer:
The book written by Jean Bodin is “Six books on the republic”.
Question 7.
Who was the proponent of internal sovereignty?
Answer:
Badin advocated Internal Sovereignty.
![]()
Question 8.
Who was the proponent of external sovereignty?
Answer:
Hugo Grotius was the proponent of external sovereignty.
Question 9.
Define Law.
Answer:
According to Holland ‘Law is a general rule of external human action enforced by sovereign political authority. ’
Question 10.
What is the root word of law?
Answer:
The term law has been derived from the Teutonic root ‘lag’, means something fixed.
Question 11.
What is the basic of moral law?
Answer:
Moral law is based on ethical code of morality of the people.
Question 12.
What is the other name of municipal law?
Answer:
National law is the other name of municipal law.
Question 13.
What is ordinance and who promulgates it?
Answer:
Ordinance are executive orders which promulgates by the president when parliament is not in session.
![]()
Question 14.
What is administrative law?
Answer:
A law which deals with civil service matters is called administrative law.
Question 15.
Who enacts laws in India?
Answer:
Parliament enacts laws in India.
Question 16.
What is meant by Liberty?
Answer:
It means the freedom of the individual.
Question 17.
What is the root word of liberty?
Answer:
The root word of liberty is the Latin word; Liber’ which means free.
![]()
Question 18.
Which revolution upheld the ideals of liberty equality and fraternity?
Answer:
France Revolution (1789) uphelds the ideals of liberty, equality, and fraternity.
Question 19.
What is Equality?
Answer:
It means equalization of opportunities.
Question 20.
What are Economic rights?
Answer: The rights enable every citizens to earn their livelihood.
Question 21.
What is meant by constitutional law.
Answer:
It is the basic law that determines the organization and functions of the government.
Question 22.
What is case law?
Answer:
Case law is a law made by courts.
Question 23.
What are the two aspects of liberty?
Answer:
Negative and Positive aspects.
Question 24.
What is natural liberty?
Answer:
It means unrestricted freedom of the individual to do what he likes.
![]()
Question 25.
What is political liberty?
Answer:
Political liberty means the right of the people to participate in the governance of the country.
Question 26.
What is National Liberty?
Answer:
It is the freedom of the nation from outside control.
Question 27.
What is a Civil Liberty?
Answer:
It is liberty available to the citizens inside society or state.
Question 28.
What is Economic Liberty?
Answer:
It is the freedom of the individual to earn his livelihood.
Question 29.
What is Natural Equality?
Answer:
It means by nature all are equal.
Question 30.
What is Civil Equality?
Answer:
It means equality before the law.
Question 31.
What is political equality?
Answer:
It means equal rights to participate in the affairs of the state.
Question 32.
What is Economic Equality?
Answer:
It means the removal of all inequalities and equal distribution of wealth.
Question 33.
What is Social Equality?
Answer:
It means there should be no discrimination on the basis of birth or family background.
Question 34.
What are Rights?
Answer:
Rights are the freedom given to individuals to do and undo certain things with status permission.
Question 35.
What is Moral Right?
Answer:
They are based on an ethical code of morality of the people. It is not enforced by state law.
![]()
Question 36.
What is duty?
Answer:
It is an obligation to do or not to do something.
Question 1.
Define sovereignty.
Answer:
According to Burgess “Sovereignty is original, absolute and unlimited power over the individual and all associations”.
Question 2.
What are the two aspects of sovereignty?
Answer:
Question 3.
Define law.
Answer:
According to Holland “Law is a general rule of external human action enforced by a sovereign political authority.
Question 4.
Mention different kinds of law.
Answer:
There are different kinds of law the important among them are: National law, International law, constitutional law, common law, ordinary law, and administrative law, etc.,
![]()
Question 5.
What is the meaning of liberty?
Answer:
Liberty’ is the power of a man to do anything that does not injure others.
Question 6.
Mention any two economic liberties.
Answer:
Question 7.
What is right?
Answer:
Right is a facility given by the legal authority7 to develop the individual personality of the citizens.
Question 8.
Mention any two political rights.
Answer:
Question 9.
What is National Law?
Answer:
It is formulated by the state legislature and it is binding on all the people within the territory of the state.
Question 10.
Write the meaning of ordinances.
Answer:
Ordinances are the executive laws which promulgates by the president to meet the unforeseen situations when parliament is not in session.
![]()
Question 11.
What is administrative law?
Answer:
A law which deals with the civil service matter and comes under administrative tribunals is called administrative law.
Question 12.
What is rule of law?
Answer:
A system in which administration carried on in accordance with the law is called rule of law.
Question 13.
What is internal sovereignty?
Answer:
A state is empower to enact, enforce and interpret the laws with in its jurisdiction is called internal sovereignty.
Question 14.
What is external sovereignty?
Answer:
A state is independent from other states and can have foreign policy with its neighbouring states is called external sovereignty.
Question 15.
Define rights.
Answer:
According to Bosanquet “A right is a claim recognised by the society and enforced by the state”.
Question 16.
Name any two political rights.
Answer:
Question 17.
What is equality?
Answer:
Facilities are provided to all the persons without any discrimination is called equality.
![]()
Question 18.
Mention the types of Liberty.
Answer:
Question 19.
Describe in brief the natural equality.
Answer:
All men are said to be born equal. This is natural equality. Human beings differ in their natural abilities and faculties.
Question 20.
What are the attributes of sovereignty?
Answer:
Absoultenegs permanence, universality, indivisibility exclusiveness originality are the attributes of sovereignty.
Question 21.
Mention types of Liberty.
Answer:
Natural Liberty, Civil Liberty, Political Liberty, Economic Liberty, National Liberty.
Question 22.
Explain Sovereignty.
Answer:
It is one of the most important elements of the sate. Bodin for the first time discussed sovereignty in the modem sense. It is the supreme power of the state.
![]()
Question 23.
How Sovereignty is absolute?
Answer:
The sovereignty of the state is absolute means it is subject to no legal limitations either internally or externally without this absolute power no state can exist.
Question 24.
What do you mean by sovereignty is indivisible?
Answer:
Indivisible means sovereignty can’t be divided. Division of sovereignty means destruction of sovereignty.
Question 25.
What do you mean by sovereignty is inalienable?
Answer:
It means sovereignty can’t be transferred. It cannot transferred to another, authority.
Question 26.
What is International Law?
Answer:
International law is a law that governs by a sovereign authority. It depends upon the acceptance and obedience of sovereign states.
Question 27.
What are the kinds of equality?
Answer:
Civil equality, Political Equality, Social Equality, National Equality, Economic Equality etc.,
Question 28.
What is Secular State?
Answer:
It is a state where there is religious freedom, and the state does not interfere with individuals’ religious matters.
Question 29.
What is legal sovereignty?
Answer:
Sovereignty according to law is known as legal sovereignty. Legal soveregin is the lawers concept of sovereignty. The authority, a person or body of persons which has power to issue and enforce the laws of state.
Question 30.
What is constitutional law?
Answer:
It is basic and fundamental law of country, it determines the organization and functions of government out in written or unwritten form.
Question 31.
What are Legal Rights?
Answer:
Legal rights are the rights which have a legal binding and are protected and recognised by state.
![]()
Question 32.
Write two fundamental duties of Indian Citizen.
Answer:
Question 33.
Mention two features of Law.
Answer:
Question 34.
What is ordinary law?
Answer:
Laws that are made by the parliament is called ordinary law.
Question 1.
Explain the features of sovereignty.
Answer:
Jean Bodin – who was the first to explain the concept of sovereignty said: “Sovereignty is the supreme power over citizens and subjects unrestrained by law.”
According to Hugo Grotius:
“Sovereignty is the supreme power vested in him whose acts are not subjected to any other who will can’t be can overridden.”
Characteristics of Sovereignty:
a. Permanent:
Sovereignty is permanent. Every state is sovereign it is accordingly permanent. The death of the rules or the change in government doesn’t mean any change in sovereign power. It comes to an end when the state is destroyed or is conquered and ruled by some external power.
b. Universality:
Sovereignty embraces each and every person and every association within the territory of the state. No individual or association in the state can disobey the sovereign authority of the state.
c. Sovereignty can’t be transferred:
The state has no right to give away its sovereignty. When a state loses or has to give up a part of the territory and population to another state, that part comes under the control of that state.
d. Indivisible:
Sovereignty can’t be divided. Division of sovereignty leads to the destruction of sovereignty.
e. Absoluteness:
Sovereignty is absolute. There can be no legal power within the state, superior to it. All individuals, associations come under the absolute power of the state. The state is completely independent.
![]()
Question 2.
Write a note on economic liberty.
Answer:
Without economic liberty, other liberty, other liberty are meaningless and useless. It means liberty of security and opportunities to find reasonable significance in the livelihood.
Question
3. Discuss kinds of equality.
Answer:
1. Natural Equality:
It implies that nature has created all men equal. It can also be defined that it insists on removing all man-made and artificial inequalities and treat all equally.
2. Civil and legal Equality:
Implies that all are equal before law and all are protected equally irrespective of caste, class, colour, race, etc.,
3. Political Equality:
Implies that all the citizens, irrespective any type of difference are entitled to participate in the affairs of state. All have equal voice in the government. It is based on principle of universal adult Franchise.
4. Economic Equality:
Implies removal of inequalities based on wealth and insists on certain minimum standard of income to all to meet their basic needs.
5. Social Equality:
Implies every individual without any discrimination must be given equal opportunity for the development of their personalities.
Question 4.
Explain the kinds of Law?
Answer
1. Natural Law:
Natural law promotes individualism and contends that there should be no law s to regulate or restrict individual behavior.
2. Constitutional law:
It is the highest and fundamental law of the land, which defines the organization of the state, determines the functions of various departments and establishes the relationship between the governor and the governed.
3. Ordinary law:
It is subordinate to the constitutional law. It is made by the parliament. It determines the relationship between the state and citizens.
4. Administrative law:
It deals with regulating the activities of the government officials in relation to state authority.
5. International Law:
International law is a body of rules and regulations which the sovereign states are expected to follow, observe and honour in their interaction with one another.
6. National Law:
National law is formulated by the sovereign authority of the State applicable to all people and associations living within territorial limits or jurisdiction of the state. It is also known as municipal law.
7. Private law:
It determines the relation between citizens.
8. Public laws:
Regulates the relation between the citizens and the states.
9. Case laws:
It refers to the laws that come into existence as a result of the decisions or verdict given by the court in a particular case. The courts give their own interpretation to apply a legal provision to a particular case. Such are known as case laws.
![]()
Question 5.
What are the kinds of Law? Explain.
Answer:
1. Natural Law:
Natural law promotes individualism and contends that there should be no laws to regulate or restrict individual behavior.
2. Constitutional law:
It is the highest and fundamental law of the land, which defines the organization of the state, determines the functions of various departments and establishes the relationship between the governor and the governed.
3. Ordinary law:
It is subordinate to the constitutional law. It is made by the parliament. It determines the relationship between the state and citizens.
4. Administrative law:
It deals with regulating the activities of the government officials in relation to state authority.
5. International Law:
International law is a body of rules and regulations which the sovereign states are expected to follow, observe and honour in their interaction with one another.
6. National Law:
National law is formulated by the sovereign authority of the state applicable to all people and associations living within territorial limits or jurisdiction of the state. It is also known as municipal law.
7. Private law:
It determines the relation between citizens.
8. Public laws:
Regulates the relation between the citizens and the states.
9. Case laws:
It refers to the laws that come into existence as a result of the decisions or verdict given by the court in a particular case. The courts give their own interpretation to apply a legal provision to a particular case. Such are known as case laws.
Question 6.
Write the relation between liberty and authority.
Answer:
It is sometimes said liberty and authority of the state as opposed to each other. But liberty doesn’t mean that unlimited right of a man to do whatever he likes. The liberty of one should not infringe on the freedom of others.
Freedom is secured through such rules and regulations. Political authority makes and enforces the law. Absolute authority will destroy liberty. Authority is the basis of liberty.
![]()
Question 7.
Explain the importance of law.
Answer:
As we live in society, we have to observe certain rules and regulations otherwise living in society will be impossible. Therefore every going out makes rules and regulations to maintain peace and order in society. The law is framed and enforced by the state to regulate the activities of citizens and to keep them within certain limits.
Question 8.
Explain the meaning of equality.
Answer:
In a literal sense, equality means that all human beings are equal. Equality doesn’t mean absolute equality. In a negative sense equality means the absence of special privileges. This means that there shall be no discriminations made on basis of caste, religion, colour, wealth, gender, etc. In a positive sense, equality means availability of adequate opportunities to all for self-development.
Question 9.
Explain characteristics of law.
Answer:
Question 10.
Write the feature of liberty.
Answer:
Question 11.
Explain what is political liberty.
Answer:
Political liberty means to be active in the affairs of the state. It can only be possible when the government is carried on in accordance with the will of the people. Political Liberty can be attained by the exercise of the four main rights.
Question12.
Write the relationship between liberty and equality.
Answer:
There are two opposite views regarding the relation between equality and liberty.
Lord Action maintained that liberty and equality are opposite to each other. But it is clear that this view is based on an interpretation of liberty as unrestricted freedom and equality as absolute equality. Hence this is wrong view.
Acc to second view, it is important to note that equality and liberty are complementary to each other. Liberty and Equality are just like two faces of same community liberty depends upon the state and laws.
![]()
Question 13.
Explain different kinds of rights.
Answer:
1. Moral Rights: are those which based on the ethical code of morality of people.
2. Legal Rights: are recognised and protected by the state.
3. civil Rights: are those rights which enable the individual to lead a normal life in society.
4. Political Rights: enable a citizen to take part in the affairs of the government.
5. Economic Rights: enable citizens to earn their living.
Question 14.
What is meant by popular sovereign?
Answer:
Question 15.
Describe two sources of law.
Answer:
Customs:
It is one of the earliest sources of law. It is the code of conduct. Habits and usage in families clans and tribes must have become well established and thus customs have been the earliest means of social regulation.
Religion:
In premitive societies custom was law was religious. Religious principles were the basis of the formation of most customs. Custom rules based on religion could not be violated. In India, Hindu law is based on the code of Manu. The Muslim law derives itself from the Quran. The influence of Christianity and the Bible in framing the laws of various western countries cannot be denied.
Question 16.
Explain negative and positive equality.
Answer:
The negative aspect, of equality, means the absence of special privileges. All barriers of birth, wealth, caste and sex should be removed in social and political life. Rights should be enjoyed – in common by all.
In its positive aspect equality means the provision of adequate opportunities for self-development to all adequate opportunities that can be given to everyone to develop the personality.
![]()
Question 17.
Distinguish between National and International law.
Answer:
National law is the branch of law that applies within the territory of the state. All persons living within the state shall obey national laws. Its violation is punishable international law is the law which regulates the relations of sovereign states with another.
International law has force only to the extent to which it is recognized and voluntarily accepted by the different states of the world.
Question 18.
What are the different kinds of liberty? Explain briefly.
Answer:
1. National Liberty:
It is associated with french thinker J.J. Rousseau. Acc to this type of man in the state of nature were completely free and there were no restrictions. This doesn’t exist in modem social life.
2. Civil liberty:
It is enjoyed by all the individuals in society. It consists of certain rights and privileges created and protected by the state.
3. Political Liberty:
This liberty is available only to the citizen either directly or indirectly participate in the political activities of the state. In short, a person makes or destroys the government.
4. Economic Liberty:
Without economic liberty, other liberty, other liberties are meaningless and useless. It means liberty of security and opportunities to find reasonable significance in the livelihood.
5. National Liberty:
This liberty implies the political independence of the state. All other liberties can’t be enjoyed unless the country is independent.
Question 19.
Write a note on the general duties of citizen.
Answer:
Duty is an obligation and a responsibility of a citizen. A citizen of a state is directed to discharge some functions and prohibited from certain functions. There are called as duties.
The duties are broadly divided into moral and legal duties.
The important general duties of a citizen are :
1. Loyalty to the state:
Every citizen should be loyal to his state. This includes his duty to defend the state during the war and his involvement in developmental activities, to protect and promote unity and integrity of the state.
2. Obedience to Law:
Every citizen should obey the laws as disobedience leads to confusion and disorder. The laws are made for the welfare of the citizens.
3. Payment of taxes:
Every citizen must honestly pay and promptly taxes for the economic development of the state.
4. Honest Exercise of the Right to Vote:
This enables citizens to elect going out of their choice which works for their development, welfare, security, protection, etc.,
Question 20.
Explain the political rights.
Answer:
1. Right to Vote:
All the citizens who have attained the age of 18 are eligible to vote through adult franchise without any discrimination.
2. Right to contest election:
All the citizens of a country who have attained a particular age are given the chance to contest elections and thus respect the aims and aspirations of various sections of society in government.
3. Right to Enter Government Service:
In a democracy, all the citizens are equally entitled to get government jobs on the basis of their qualifications.
4. Right to petition:
It gives an opportunity to all people to bring their problems to the notice of the government and seek remedies.
5. Right to Criticism:
It is one of the biggest boons of democracy. All citizens have the right to criticise the policies of the government.
Question 21.
What is the importance of liberty?
Answer:
1. Respect for individuals:
Liberty is essential for every individual to be at his best self, subject, to legal limitations.
2. Respect for laws:
Liberty is required to put down any kind of dominance within a civil society where all enjoy equal liberty and invasion of liberty of a person is nothing but a violation of liberty which is visited by penalty or punishment.
3.Respect for fellow being:
The fear of punishment in case of violation of the liberty of others; makes one respect the feelings of others thereby ensuring respect for fellow
beings.
4. Promotes co-operation and understanding:
Liberty ensures individuals to develop harmony, brotherhood, and understanding based on mutual necessity and co-operation.
![]()
Question 22.
Answer:
Explain the importance of Equality.
1. Self-respect:
In the absence of discrimination, all can develop as they wish, thus ensuring respect for individual personality.
2. Promotes Co-operation:
When all are treated equal in the eyes of law, people can lead a life based on cooperation and understanding, develop harmony, which will lead to the brotherhood.
3. Prevents revolution:
When all are treated equal, they can pursue what they consider best and it would prevent revolutionary tendencies from taking over the system.
Question 23.
Explain the importance of rights.
Answer:
1. Development of personality: Rights describe the do’s and don’ts of civilized life and this helps an individual to develop his personality.
2. Symbol of national prestige: The quality Of rights in a country symbolizes the quality of its rules and people.
3. Respect for fellow beings: Rights guarantees respect for fellow citizens.
4. Protection against government: Rights guarantees protection against government action.
Question 24.
Explain the importance of Sovereignty?
Answer:
1. Identifies the nation-state: Sovereignty has contributed to the identity and status of the nation-state.
2. Sovereignty gives freedom to the state: Sovereignty increases the freedom of nation-state, so as to decide on its domestic and foreign policy.
3. Allows progress: Sovereignty provides motivation for each nation to achieve progress.
4. Promotes nationalism: Sovereignty promotes national spirit and allows love for the nation.
5. Allows the state to exercise coercive power: Sovereignty monopolies the state internally and externally.
Question 1.
What is the law? explain its kinds.
Answer:
1. Natural Law:
Natural law promotes individualism and contends that there should be no laws to regulate or restrict individual behavior.
2. Constitutional law:
It is the highest and fundamental law of the land, which defines the organization of the state, determines the functions of various departments and establishes the relationship between the governor and the governed.
3. Ordinary law:
It is subordinate to the constitutional law. It is made by the parliament. It determines the relationship between the state and citizens.
4. Administrative law:
It deals with regulating the activities of the government officials in relation to state authority.
5. International Law:
International law is a body of rules and regulations which the sovereign states are expected to follow, observe and honour in their interaction with one another.
6. National Law:
National law is formulated by the sovereign authority of the state applicable to all people and associations living within territorial limits or jurisdiction of the state. It is also known as municipal law.
7. Private law:
It determines the relation between citizens.
8. Public laws:
Regulates the relation between the citizens and the states.
9. Case laws:
It refers to the laws that come into existence as a result of the decisions or verdict given by the court in a particular case. The courts give their own interpretation to apply a legal provision to a particular case. Such are known as case laws.
![]()
Question 2.
Explain the meaning and kinds of rights.
Answer:
1. Moral Rights: are those based on the ethical code of morality of people.
2. Legal Rights: are recognised and protected by the state.
3. civil Rights: are those rights that enable the individual to lead a normal life in society.
4. Political Rights: enable a citizen to take part in the affairs of the government.
5. Economic Rights: enable citizens to earn their living.
Question 3.
Explain the meaning and kinds of liberty.
Answer:
1. National Liberty:
It is associated with french thinker J.J. Rousseau. Acc to this type of men in the state of nature were completely free and there were no restrictions. This doesn’t exist in modern social life.
2. Civil liberty:
It is enjoyed by all the individuals in society. It consists of certain rights and privileges created and protected by the state.
3. Political Liberty:
This liberty is available only to the citizen either directly or indirectly participate in the political activities of the state. In short, a person makes or destroys the government.
4. Economic Liberty:
Without economic liberty, other liberty, other liberty are maningless and useless. It; means liberty of security and opportunities to find reasonable significance in livelihood.
5. National Liberty:
This liberty implies the political independence of the state. All other liberties can’t be enjoyed unless the country is indepent.
Question 4.
Describe the political rights.
Answer:
a. Right to Vote:
All the citizens who have attained the age of 18 are eligible to vote through adult franchise without any discrimination.
b. Right to contest election:
All the citizens of a country who have attained a particular age are given the chance to contest elections and thus respect the aims and aspirations of various sections of society in government.
c. Right to Enter Government Service:
In a democracy, all the citizens are equally entitled to get government jobs on the basis of their qualifications.
d. Right to petition:
It gives an opportunity to all people to bring their problems to the notice of the government and seek remedies.
e. Right to Criticism:
It is one of the bigget boon of democracy. All citizens have the right to criticise the policies of the government.
![]()
Question 5.
Explain economic rights.
Answer:
The rights which enable every citizens to earn their livelihood are called economic rights.
Following are the important economic rights.
Question 6.
Distinguish between legal sovereignty and political sovereignty.
Answer:
A legal sovereign is a person or body of persons in a state which is the supreme law-making authority disobedience to law result in punishment.
The characteristics are :
The legal sovereign simply makes laws. The wishes of political sovereign who can’t express the will of the state. It is not so easy to define political sovereign. It is unorganized and unknown to law. Political sovereign and legal sovereign concide in direct democracy.
Dejure and Defacts Sovereignty.
Dejure:
Sovereignty means sovereignty by law. In Other words, dejure is the lawful and legitimate sovereign. It is legally constituted and has the basis and authority of law. It has legal right to govern.
Defacto:
Sovereignty is which has no legal basis because it is not recognized by law but exists on fact. Defacto comes to power usually by force. If it manages to stick to power and show that he can rule he becomes dejure sovereign.
Titular Sovereignty and Real Sovereignty.
Titular has the power but he won’t use it but somebody else will use. For eg: King or Queen of England and the President of India. The council of ministers is the real soveregin and occupy a highly dignified and privileged position in the state. In a presidential system, there is no difference because the president himself exercises his powers and functions.
Question 7.
Define Law. Explain its features.
Answer:
The word law is derived from the Teutonic word Tag’ which means something fixed.
Acc to John Austin: “a command given by a superior to an inferior.”
Accordingly to Woodrow Wilson “Law is that position of established thought and habit which . has gained distinct and formal recognition in the shape of uniform rules.”
Characteristics of law:
Question 8.
Explain Civil Rights.
Answer:
1. Right to live:
All people born in society have the right to live. The main responsibility of the state is to provide security of life to the people.
2. Right to Liberty:
All the people living in the state have the right to travel freely all over the country. They can reside anywhere as they desire.
3. Right to Religion:
All the citizens have the freedom to practice or propagate any religion they want.
4. Right to Culture and Education:
All the citizens have the right to protect, develop and practice their own culture. The right to education has been granted, to enable a person to develop and improve his or her personality.
5. Right to Association:|
All the citizens have the freedom to form the different types of association as per their needs and aspirations.
6. Right to Family:
Family is an integral part of human existence. Every individual has the right to marriage, to bring up children and to continue his generation.
![]()
Question 9.
What are the various sources of law?
Answer:
1. Customs:
Customs is one of the earliest sources of law. It is the code of conduct observed by people in society. When the state was established, these customs received
recognition and became laws.
2. Religion:
In many countries, religion is the basis of the law and it lies as the root of many national legal systems.
3. Equity:
It refers to the principles of equality, fairness, and justice in which judges apply. When existing laws provide no relief in a particular case, judges apply the principle of equity.
4. Judicial decisions:
Judges interpret mould and extend the existing laws. Such decisions of the judges later become precedents judicial precedents supplement the law and are applied in the future cases of a similar kind.
5. Scientific commentaries:
These are scientific discussions by eminent jurists or lawyers. It is an important source of material for lawmakers.
6. Legislation:
It means the enactment and promulgation of laws by the legislature. It forms an important source of law.
You can Download Chapter 1 Political Science as a Discipline Questions and Answers, Notes, 1st PUC Political Science Question Bank with Answers Karnataka State Board Solutions help you to revise complete Syllabus and score more marks in your examinations.
Question 1.
Who is known as the Father of Political Science?
Answer:
Aristotle is known as the father of Political Science.
Question 2.
Who studied political science first scientifically?
Answer:
Aristotle studies political science first scientifically.
Question 3.
Name the book written by Aristotle.
Answer:
Aristotle wrote the book “The Politics”.
![]()
Question 4.
Name any one of the city-state of ancient Greece?
Answer:
Sparta is one city-state of ancient Greece.
Question 5.
What is political science?
Answer:
A study of the political activities of human being is called political science,
Question 6.
Who called political science as supreme or master science?
Answer:
Aristotle.
Question 7.
Who said, ‘man is a social animal’?
Answer:
Aristotle.
Question 8.
How many constitutions did Aristotle study prior to writing ‘The politics’?
Answer:
Aristotle studies 158 constitutions prior to write ‘The politics’.
Question 9.
Which is the root word of Politics’?
Answer:
Politics is derived from the Greek word ‘polis’.
![]()
Question 10.
What is the meaning of word ‘polis’?
Answer:
‘Polis’ means Greek City-States.
Question 11.
Name the book written by Machiavelli?
Answer:
The book written by Machiavelli is ‘The prince’.
Question 12.
Which is the root word of Political Science?
Answer:
Greek word‘Polis’is the root word of Political Science.
Question 13.
To which branch of science does Political Science belong?
Answer:
Political Science is an exact science. It belongs to the category of Social Science.
![]()
Question 14.
Which scholar considered Political Science to be both normative and descriptive?
Answer:
MAX WEBER considered Political Science to be both normative and descriptive.
Question 15.
What are the major divisions of Political Science?
Answer:
The major division of Political Science is
Question 16.
Mention the writers who believed Political Science to be a study of the state alone.
Answer:
Garies, Garner, Goodnow, and Bluntschli believed political science to be the study of the state alone.
Question 17.
For what reason Political Science cannot be regarded as science?
Answer:
![]()
Question 18.
Who said a man is a social animal?
Answer:
Aristotle has said man is a social animal.
Question 19.
Write the definition of Political Science given by G W. Garner.
Answer:
G W. Garner: Political science begins and ends with the state.
Question 20.
Who said political Science is a master science?
Answer:
Aristotle said political science is a master science.
Question 21.
Who wrote the book ‘Republic’?
Answer:
Plato wrote the book ‘Republic’.
Question 22.
What is the subject matter of Political Science?
Answer:
State and Government are subject matter of Political Science.
![]()
Question 23.
What is the meaning of ‘Politics’?
Answer:
The word ‘Politics’ is derived from the word ‘Polis’, which means city to Greeks, which deals with city-state.
Question 1.
Define political science.
Answer:
Political Science is the part of social science which deals with the study of the state, government, law, people, power and other political concepts of the state. Aristotle considered the father of political science.
Question 2.
Name the divisions of the scope of political science.
Answer:
Question 3.
What is the subject matter of political science?
Answer:
The subject matter of political science is state and government.
![]()
Question 4.
Why is ‘man is a social animal’?
Answer:
Aristotle said, man, is a social animal because man can fulfill his basic desires, needs, requirements and protect himself. He cannot live all alone himself and he is not self-sufficient and self reliant.
Question 5.
What is the scope of political science?
Answer:
The scope of political science means boundary or the sphere of the province of the subject. It means the subject matter of the study.
Question 6.
What do you know about the Greek city-states?
Answer:
In the ancient days, the Greeks lived in city-states. In those days, city-states were very small. People were living in small city-states and direct democracy was existing in those city-states.
Question 7.
In which of the ancient Indian work do we find reference to state administration?
Answer:
We find references of state administration in the works of Kautilya in ‘Arthashastra’, Kamandaka in ‘Neetisara’, Rigveda, Yajurveda, Shukraneetisara and Manusmrithi.
Question 8.
When and where was the International Political Science Conference held?
Answer:
The International Political Science Conference was held in Paris in 1948 under the UNESCO conference.
Question 9.
Who is called the Greek Philosopher Trio?
Answer:
Socrates, Plato, and Aristotle are called a Greek philosopher Trio.
![]()
Question 10.
Who are the writers who said political science is a science?
Answer:
Aristotle, Bodin, Hobbes, Machiavelli, Montesque, Lewis, Bluntschli argues that political science is a science.
Question 11.
Who are the writers who said political science is an art?
Answer:
J.S. Mill, Buckle, August Compte, Maitland, James Bryce, and Charles A. Bryde says that political science is an art.
Question 12.
Write the definition of Political Science given by Paul Janet.
Answer:
According to Paul Janet, “Political Science is’ that part of social science, which treats the foundation of the state and principle of government”.
Question 13.
Explain the nature of political Science.
Answer:
He who is unable to live in society must be either a beast or a god. It means that man is a social animal. He cannot lead a life of isolation. He has to live in society. To satisfy his wants man has to live in society. Man is not only a social animal but he also a political animal. He is quarrelsome by nature, to control this behaviour some laws are needed.
Question 14.
Is Political Science a Philosophy?
Answer:
Some writers consider political science as a part of philosophy because it deals with abstract concepts. But we cannot fully accept this argument because political science deals not only .with theories and ideas but also with practice.
![]()
Question 15.
Is the study of Political Science a must for every Politician?
Answer:
Politician is a person who takes an active part in politics. It is very much necessary for a politician to study political science. It teaches him good governance about the state and working of the government.
16. Is the study of political Science necessary for every citizen?
Answer:
The study of political science is very much necessary for every citizen, especially in a democracy. To know the value of vote, rights and to take part actively in the administration of the government.
Question 17.
Write the two definitions of Political Science.
Answer:
According to Paul Janet “Political Science is that part of social science which treats the foundation of the state and principles of government”. According to Gilchrist “Political Science deals with state and government”.
Question 18.
Who are the writers who said Political Science is the study of the state alone?
Answer:
Garies, Garner, Goodnow, and Bluntschli are a group of thinkers who maintain that only the state is the subject matter of political science.
Question 19.
Who are the writers who say political science is the study of government alone?
Answer:
Stephen Leacock, John Seelay and Robson argued that political science deals with the only government. They believed that the subject matter of political science is the government.
Question 20.
Mari cannot live in isolation, Why?
Answer:
Man cannot live in isolation because he is a social animal and as such he is born and lives in the society living together give protection and security and man can fulfill all his basic necessities.
![]()
Question 21.
Give Gettell’s definition of political science.
Answer:
According to Gettell, Political Science is a historical investigation of what the state has been, an analytical study of what the state is and a political ethical discussion of what the state should be.
Question 22.
Mention any one major work of Plato and Aristotle.
Answer:
Plato – The Republic. Aristotle – Politics.
Question 1.
Discuss the evolution of political science.
Answer:
Man can understand the world with the help of science. Science is classified into different divisions such as physical science, biological science, and social science. Physical science helps us to discover new innovations, Biological science helps us to know nature. Social science helps us to know- the world in different dimensions. Social science includes history, economics, sociology, political science, geography, psychology, Ethics, philosophy, etc. Political science is one of the important branches of social science and occupies a very significant role in understanding the evolution of human beings.
The subject matter of political science is an ancient one. The ancient Greek philosophers have laid a good foundation for political science. In fact, the ancient Indian thinkers have referred to the concepts of political science in their works and historical documents. References are made on the state administration in the works of Kautilya in ‘Arthashatra1, Shukraneetisara of Shukra and Manusmirthi of Manu. But these works are focused more on religious consciousness rather than political awareness.
Hence the study of political science was neither systematic nor scientific during that period. The credit for developing the theory of political science on a systematic and scientific basis rightly goes to the Greeks. Socrates a great ancient Greek philosopher contributed his political thinking through dialogues.
Plato emphasized his political views in his work ‘The Republic’. Whereas, Aristotle went a step ahead and made his approach to the study of political science more realistic. He laid a scientific foundation for the study of political science and regarded it as the “Supreme science”. He expressed his political thinking in his familiar work “The Politics”. In the modem context, Machiavelli of Italy added a new dimension to the study of political science in his great work “The prince”.
![]()
Question 2.
Explain the nature of political science.
Answer:
Aristotle says that man by nature is a social animal. He born in society, live in society and die in society only. To fulfill his needs, he created society. In society only he can enjoy all the facility’s love and affection. Away from society he maybe God or ghost. Man is a social being at the same time he is a political being also.
He is a selfish, egoist and quarrelsome by nature. The attitudes of selfish, egoist and jealousy of a man lead to anarchy in the society while leading the life. So the order of the society may be disrupted and man cannot lead his life happily and peacefully.
In order to control the bad behavior of such people and establish a peaceful society, there should be rules and regulations. The state has emerged to frame and implement these rules through its agency “The Government”. So the state controls the political activities of the human being and restore peace in the society. The subject which studies about the state, government, the political activities of a human being is regarded as political science.
Question 3.
Explain the scope of political science.
Answer:
Aristotle described political science as a ‘master science’ which made it perhaps the greatest contribution to the making of political science scientific. Hie term “Scope” refers to the subject or the boundaries of a particular branch of knowledge. There is no perfect agreement among the political thinkers as to the problems, which come under the study of political science. Broadly speaking, there are three groups of writers holding different views on the scope of political science.
The first group of writers like Garies, Gamer, Goodnow, and Bluntschli restricted the scope of political science only to the study of the state.
The second group of writers like Prof. Sheley and Dr. Stephen Leacock said that political science deals with government only.
The third group of writers like Gettell, Gilchrist, Paul Janet, and Prof. Laski maintained that the scope of political science extends to both state and government. Prof. Laski maintains that the state, in reality, means the government.
We may agree with the third group of writers that political science is a study of both state and government is the steering wheel of the ship of the state. There can be no state without a government, the state remains the central subject of our study, and the whole mechanism of government revolves around it.
Scope according to the UNESCO; the international Political Science Association at its Paris Conference in 1948 discussed the scope of political science and marked out its subject matter as follows:
1. Political Theory:
Political Theory, History of Political Ideas.
2. Government:
The Constitution, the Government-Regional and Local Government, Public Administration, Economics and Social functions of government, Comparative political institutions.
3. Parties, Groups and Public Opinion:
Political Parties, Group and Associations, Citizen Participation in Government and administration, Public Opinion.
4. International Relations:
International relations, International organization, and Administration, International Law.
Question 4.
Is political science a Science? Explain.
Answer:
Aristotle called political science as supreme or Master science. Many writers like Thomas Hobbes, Jean Bodin, and Lord Bryce agree that political science is a science.
Reasons for it to be called as a science are:
1. Political Science is a systematic study:
Just like the science subjects which are studied systematically, in the same way. it is possible to make a study of political science. There is a connection between cause and effects.
2. Experiments are possible in Political Science:
Politics is also a systematized knowledge that has been acquired by systematic observation, experiment and study For e.g. framing the Indian constitution and the methods of direct democratic devices adopted in Switzerland.
3. Common principles, conclusions possible:
Political scientists have arrived at broad principles after observing the political phenomenon over a long period of time. Reasons for the rise of new states, the eruption of revolts and revolutions, the emergence of political parties, etc, have been discovered.
4. Relationship between the causes and effects can be known:
In political science with a greater degree of approximation, the relationship between causes and effects can be established.
5. More exact than other social sciences:
Political science has the status of being more exact than other social sciences.
Question 5.
Is Political science is an art? Explain.
Answer:
1. Absence of Uniform terminology:
The study is called by different terms and there is no unanimity among thinkers regarding the exact term. The subject is called politics, political science, political theory, political philosophy, applied politics, theoretical politics, etc.
2. Differences regarding methods, definition, and scope:
There is no single method of studying the subject but different methods are available. Political principles are not universally accepted and applied. Terms like democracy, socialism, communism, liberalism, and Nationalism have been defined and explained in different ways.
3. Absence of Precision:
Political science lacks precision in the study of state and government or theories like democracy.
4. Experimentation is not possible:
Conducting experiments in political science is difficult since it deals with human beings and their behavior is unpredictable.
5. Absence of Universal Laws:
Political science has certain concepts acceptable with similar meaning but there is no universal truth.
6. Difficult to predict the future:
Political researchers find it difficult to predict the future course of events since political events are subject to ever-changing public opinion.
7. Lack of Objectivity:
The political scientists cannot be objective since he is influenced by race, religious, economic and other considerations.
Question 6.
Explain the origin and development of political science.
Answer:
According to Aristotle, political activity can be traced to the very nature of man. It is true that no individual can exist or live in isolation. No one is self-sufficient. So, man is a social animal. In the same manner, he is a political animal. Hence society requires an organization to regulate and control human activities. The organization states, which in turn function through its agency, government.
Question 1.
Explain the Nature and Scope of Political science.
Answer:
Aristotle described political science as a ‘master science‘ which made it perhaps the greatest contribution to the making of political science scientific. The term “Scope” refers to the subject or the boundaries of a particular branch of knowledge. There is no perfect agreement among the political thinkers as to the problems, which come under the study of political science.
Broadly speaking, there are three groups of writers holding different views on the scope of political science.
The first group of writers like Garies, Gamer, Goodnow, and Bluntschli restricted the scope of political science only to the study of the state.
The second group of writers like Prof. Sheley and Dr. Stephen Leacock said that political science deals with the government only.
The third group of writers like Gettell, Gilchrist, Paul Janet, and Prof. Laski maintained that the scope of political science extends to both state and government. Prof. Laski maintains that the state, in reality, means the government.
We may agree with the third group of writers that political science is a study of both state and government is the steering wheel of the ship of the state. There can be no state without a government, the state remains the central subject of our study, and the whole mechanism of government revolves around it.
Scope according to the UNESCO; the international Political Science Association at its Paris Conference in 1948 discussed the scope of political science and marked out its subject matter as follows:
Political Theory:
Political Theory. History of Political Ideas.
Government:
The Constitution, the Government-Regional and Local Government, Public Administration, Economics and Social functions of government, Comparative political institutions.
Parties, Groups and Public Opinion:
Political Parties, Group and Associations, Citizen Participation in Government and administration, Public Opinion.
International Relations:
International relations. International organization and Administration, International Law.
![]()
Question 2.
Explain the relevance of the study of political science in the contemporary world.
Answer:
The study of political science is very useful to individuals and society. As said by Bernard Shaw that “political science by which alone civilization can be saved”. Political science exerts its influence on the life and destiny of human beings. Why political science has become so important in the life of man? It can be judged on the following grounds:
1. Essential for a cultured existence:
The study of Political Science introduces an individual to the requirements of a civilized and cultured society.
2. To understand the evolution of state and government:
Political Science is a study of state and government in all its aspects, in order to understand the roots of state and government such as how and why the state and government came into existence and .why it continues to be in existence and what are the essential factors that make a state, the types and forms of government, the aims and objectives of the government, etc., the knowledge of political science is essential.
3. To provide knowledge of rights and duties:
Every citizen, especially in a democracy desires to have knowledge of rights and duties. It is in political science that one learns all the aspects of these rights and duties.
4. To make Democracy effective:
Democracy today is the most popular system of government. Many factors contribute to the success and effectiveness of democracy. Political science enables one to understand all the principles, merits and defects of democracy.
5. Provides political education:
The study of political science provides political education to a citizen so that he may understand national problems in a new perspective.
6. Provides knowledge of ideologies:
The study of political science provides a vast knowledge of various prevailing ideologies like democracy, socialism, communism, nazism, fascism, etc.
7. Knowledge useful to political leaders and bureaucrats:
Knowledge of political science is very useful to the political leaders to carry on the working of the government effectively. Knowledge of the constitution is necessary for everyone to contribute better to the functioning of the government.
8. To understand political power:
Political Science helps citizens to understand the concept of political power; how governments function; what are the interest and forces behind the policies; who are his elected representatives and what they stand for.
9. To promote human warfare:
Political Science in the contemporary’ period lies the fact that it creates conditions for economic progress since it is motivated by an interest in human welfare.
10. To understand international relations and contribute to peace:
International relations is one of the specialized branches of political science dealing with the relationship among sovereign states. It includes many concepts like balance power, collective security alliances, treaties, international law, international organizations, etc.
![]()
Question 3.
Is political science a Science or an art? Explain.
Answer:
Aristotle called political science as supreme or Master science. Many writers like Thomas Hobbes, Jean Bodin, and Lord Bryce agree that political science is a science.
Reasons for it to be called as a science are:
1. Political Science is a systematic study:
Just like the science subjects which are studied systematically, in the same way, it is possible to make a study of political science. There is a connection between cause and effects.
2. Experiments are possible in Political Science:
Politics is also a systematized knowledge which has been acquired by systematic observation, experiment and study. For e.g. framing the Indian constitution and the methods of direct democratic devices adopted in Switzerland.
3. Common principles, conclusions possible:
Political scientists have arrived at broad principles after observing the political phenomenon over a long period of time. Reasons for the rise of new states, the eruption of revolts and revolutions, the emergence of political parties, etc, have been discovered.
4. Relationship between the causes and effects can be known:
In political science with a greater degree of approximation, the relationship between causes and effects can be established.
5. More exact than other social sciences:
Political science has the status of being more exact than other social sciences. Some writers consider political science as a part of political philosophy. It may be noticed that every political theory owes its origin to some philosophical idea. But however, we need not accept this argument in political science, not only deal with theories and ideas but also with practice.
In political science, there is an attempt to apply theoretical knowledge to the practice of politics. An art to be perfected require intelligence and constant practice by artist. On the basis of the above discussions, it may be concluded that it is an art.
1. Absence of Uniform terminology:
The study is called by different terms and there is no unanimity among thinkers regarding the exact term. The subject is called politics, political science, political theory, political philosophy, applied politics, theoretical politics, etc.
2. Differences regarding methods, definition, and scope:
There is no single method of studying the subject but different methods are available. Political principles are not universally accepted and applied. Terms like democracy, socialism, communism, liberalism, and Nationalism have been defined and explained in different ways.
3. Absence of Precision:
Political science lacks precision in the study of state and government or theories like democracy.
4. Experimentation is not possible:
Conducting experiments in political science is difficult since it deals with human beings and their behavior is unpredictable.
5. Absence of Universal Laws:
Political science has certain concepts acceptable with similar meaning but there is no universal truth.
6. Difficult to predict the future:
Political researchers find it difficult to predict the future course of events since political events are subject to ever-changing public opinion.
7. Lack of Objectivity:
The political scientist cannot be objective since he is influenced by race, religious, economic and other considerations.
Question 4.
Define political science. What is the significance or utility of its study?
Answer:
The term ‘Politics’ was used by the great Greek Philosopher Aristotle. It is derived from the term ‘Polis’ which means ‘city’ to Greeks city was the state and subject that was dealing with city-state was called politics. So political science means a systematic study of the state and government.
The following are some of the important definitions.
Political science has become the most important branch of social science. Aristotle called it a master science. Democracy has become the most common form of government nowadays. The study of political science trains the people for the successful working of democracy. The study of political science enables the individual to understand his rights and duties and enlightens him to take an active part in the affairs of the state.
It also promotes a sense of responsibility and the spirit of patriotism.
The citizen can understand the nature, merits, and demerits of different types of Government and other administrative institutions in their state by studying political science. Studying political science will enable people to understand different theories and ideologies and help him to decide whether to accept it or reject it.
The study of political science enables to understand international relations and international law. It helps to understand thoroughly the significance of world organizations in solving international problems and in maintaining international people.
Students can Download Karnataka Class 10 Hindi निबन्ध-लेखन, KSEEB SSLC Class 10 Hindi Solutions helps you to revise the complete Karnataka State Board Syllabus and score more marks in your examinations.
आ) निबन्ध-1:
Population Problem Summary in English:
Introduction to the matter: Population Problems is the problem of the world. There will be a birth of two babies for every second in this world. But in India, this problem has become the most burning issue. So for as area is concerned India stands in the 7th place. Amongst all the countries on this globe. But in Population, it stands in the second place. It is just one place below China, that stands first in Population. At present India has 100 crores of Population.
Reasons for population growth :
The growth of the population may lead to many types of crimes. These crimes spoil the environment and the spoiled environment generates diseases. It results in an unhealthy society.
Nowadays, both central and state governments are planning a number of welfare plans for women’s and even in schools, ladies are taking education free of cost. So women must understand their responsibility and help the nation in controlling the Population. Everyone must understand that our family should have only two children so that our family will be happy.






निबन्ध – 2:
Women Part Of Sincerity Summary in English:
There is a respectable place for ladies in India. Since ancient days women have been respected with trust and devotion. The woman is in the form of a mother, sister, wife and in many other forms. A man’s life is incomplete without a woman. There is a good wherever a woman is respected. Since the time of Veda and Upanishad, a woman has learnt different Shastras mathematics and science.
Our Indian rivers have been given women names like Ganga, Yamuna, Kaveri and Narmada. Thus a woman has been given maximum importance. But liberty has been taken away from her. A woman is under the shelter of her father in her childhood days and under the control of her husband and her children lastly. Now the situation has been changed and a woman is becoming equal to man in each and every field like science, education music, employment and politics. E.g. Sarojini Naidu became the first woman governor and showed Gandhi became the Primeminister of her India and showed her guts to the world.


निबन्ध – अभ्यास
निबन्धों की रूपरेखा
I. पर्यटन का महत्व:
II. नागरिक के कर्तव्य
III. बेरोजगारी
IV. महँगाई:
V. वन महोत्सव:
भ्रष्टाचार की समस्या।
भ्रष्टाचार का अर्थ है – अनैतिक अथवा अनुचित व्यवहार। आज देश में चारों ओर भ्रष्टाचार फैला हुआ है। यदि समय रहते इस पर नियंत्रण नहीं किया गया तो सारे देश का वातावरण बिगड़ सकता है। मनुष्य बड़ा लालची है। वह बहुत कुछ पाने के लिए अनैतिक मार्ग अपनाता है। इस प्रकार भ्रष्टाचार बढ़ता जाता है। भाषावाद, क्षेत्रीयता, जातीयता आदि से भी भ्रष्टाचार बढ़ता है।
भ्रष्टाचार के अनेक रूप हैं। चोरबाजारी, रिश्वतखोरी, दल-बदल, जोर-जबरदस्ती – ये सब भ्रष्टाचार के ही रूप हैं। धर्म का नाम लेकर लोग अधर्म का काम करते हैं। अधिकार, कुर्सी के लिए भी भ्रष्टाचार पनपता है। परिणामतः समाज में भय, आक्रोश व चिंता का वातावरण बनता है। यह हमारी व्यवस्था को बिगाड़ता है। भ्रष्टाचार विकास की प्रगति में बाधा है। आजकल सेना में भी इसकी चर्चा सुनी गई है जो हमारे लिए चिंता का विषय है।
भ्रष्टाचार किसी एक व्यक्ति की नहीं, बल्कि संपूर्ण समाज और देश की समस्या है। इनका पूर्णविनाश सामूहिक प्रयास से ही संभव है। इसके लिए शासन की इच्छा शक्ति होना जरूरी है। भ्रष्टाचार के आरोपियों को कठोर से कठोर दंड देना चाहिए। भ्रष्टाचार को मिटाने के लिए सरकार, समाज और प्रत्येक नागरिक को मिलकर कार्य करना चाहिए। लगता है, हमारी शिक्षण प्रणाली में कई कमियाँ हैं, सामाजिक नैतिकता की कमी होना अच्छा नहीं है। आइए, हम संकल्प करें कि न हम रिश्वत देंगे और न रिश्वत लेंगे।
दहेज प्रथा।
हम प्रतिदिन समाचार-पत्रों में पढ़ते हैं कि दहेज के कारण अमुक युवती की हत्या की गई, अमुक लड़की ने आत्महत्या कर ली, अमुक स्त्री को प्रताडित किया गया और अमुक महिला को घर से निकाल दिया गया। वास्तव में दहेज प्रथा हमारे समाज के लिए और हमारे देश के लिए एक बहुत-बड़ा कलंक है।
माता-पिता द्वारा अपनी पुत्री को इच्छानुसार जो वस्तुएँ, धन-संपत्ति विवाह के समय दी जाती थी, वही दहेज माना जाता था। लेकिन इस पद्धति में परिवर्तन होते गए। विवाह से पूर्व ही वर-पक्ष की ओर से दहेज के रूप में कई माँगें रखी जाने लगी। परिणाम-स्वरूप समाज में कई विसंगतियाँ शुरू हो गई।
दहेज के कारण प्रायः लड़कियों को ही कष्ट भोगना पड़ता है। यह पद्धति अमीर, गरीब सभी में देखी जा रही है। अमीर तो जैसे-तैसे दे देगा, परन्तु गरीब क्या देगा? फिर यहीं से शुरू होता है अत्याचार। नवविवाहितों की प्रताड़ना, उनकी हत्या, उन्हें जलाना जैसे अमानवीय कृत्य बढ़ जाते हैं।
यद्यपि दहेज-प्रथा के विरोध में कई कानून बने हुए हैं, कई सामाजिक संस्थाओं द्वारा भी दहेज न लेने के लिए प्रचार किया जाता है; फिर भी यह कलंक अभी मिटा नहीं है। वर-वधु दोनों पक्षों को चाहिए कि वे न दहेज लें और न दें। वास्तव में समाज में इसके लिए जागृति होनी चाहिए। हमें उनका बहिष्कार करना चाहिए जो दहेज लेते या माँगते हैं। तभी हम दहेज-विहीन समाज का निर्माण कर पायेंगे।
स्वतंत्रता-दिवस।
स्वतंत्रता-दिवस अर्थात् 15 अगस्त हमारे देश के इतिहास का यह स्वर्णिम-दिन है। इसी दिन हमारा देश अंग्रेजों की गुलामी से छुटकारा पाकर स्वतंत्र हुआ था। लालकिले पर देश के प्रथम प्रधानमंत्री पं. नेहरू जी ने तिरंगा झंडा फहराया था।
देश की आजादी के लिए भारतीयों ने एक लम्बी लड़ाई लड़ी थी। हजारों-लाखों के बलिदान के पश्चात् हमें यह आजादी मिली है। इस स्वतंत्रता के लिए 1857 को सबसे पहले क्रांति हुई थी। उसी से प्रेरणा लेकर, आगे लोकमान्य तिलक, दादाभाई नौरोजी, गोपालकृष्ण गोखले, लाला लाजपतराय, महात्मा गाँधी जैसे अनेकानेक देशभक्तों ने 1947 तक आन्दोलन किये।
यद्यपि कुछ हद तक गाँधीजी के नेतृत्व में यह सत्य और अहिंसा की लड़ाई थी, फिर भी आजादी की लड़ाई में बहुत-बड़ी संख्या में देशभक्त शहीद हुए हैं। इस बात को हमें नहीं भूलना चाहिए। इस लड़ाई के संदर्भ में अंग्रेजों ने भारतीयों पर बहुत-बड़े अत्याचार किए थे, लाठियाँ बरसाई, देशभक्तों को जेलों में दूंस दिया और गोलियाँ भी बरसाई गई। अंत में अंग्रेजों को हार माननी पड़ी और वे 15 अगस्त 1947 को अपना बोरिया-बिस्तर समेटकर चले गए।
तब से प्रतिवर्ष 15 अगस्त को सम्पूर्ण देश में स्वतंत्रता दिवस मनाया जाता है। देश के प्रधानमंत्री दिल्ली में राष्ट्रध्वज फहराते हैं। नगर-नगर, गाँव-गाँव में प्रभात-फेरियाँ निकलती हैं। देशभक्ति के गीतों की गूंज हर कहीं सुनाई देती हैं। नेताओं के भाषण होते हैं। सांस्कृतिक कार्यक्रमों के आयोजन होते हैं। बलिदानियों को स्मरण कर, उन्हें श्रद्धांजलि दी जाती है। हमारा कर्तव्य है कि हम इस आजादी को बनाये रखें। देश-द्रोहियों को करारा जवाब दें।
पुस्तकालय।
मनुष्य के जीवन में ज्ञान का बड़ा महत्व है। ज्ञान-प्राप्ति केवल स्कूल-कॉलेजों में ही नहीं, बल्कि पुस्तकालय में भी होती है। सचमुच पुस्तकालय पवित्र तीर्थस्थान है। यह एक सरस्वती का पावन मंदिर है।
पुस्तकालय में साहित्य, विज्ञान, इतिहास, राजनीति, कला, दर्शनशास्त्र आदि अनेक विषयों की पुस्तकें होती हैं। उनमें कुछ संदर्भ ग्रंथ और शब्दकोश भी होते हैं। धनवान भी सभी विषयों की पुस्तकें नहीं खरीद सकता। समृद्ध पुस्तकालयों में दैनिक व साप्ताहिक समाचार-पत्र भी मंगाए जाते हैं।
कोई भी व्यक्ति ऐसे सार्वजनिक पुस्तकालय से लाभ उठा सकता है। पुस्तकालय का सदस्य बन सकता है। पुस्तकें पढ़ने के लिए अपने घर भी ले जा सकता है। इस प्रकार मनुष्य पुस्तकालयों से ज्ञान-सागर में डुबकी लगा सकता है।
जब भी समय मिले, तो पुस्तकालय में जाकर समय बिताना लाभकारी है। महान लेखकों की पुस्तकें पढ़ने और मनोरंजन करने, ज्ञान-विकास के लिए पुस्तकालय बड़े उपयोगी हैं। देश-विदेश का साहित्य पढ़ने से लोगों में जागृति होती है। पुस्तकालय समाज और देश के लिए हितकारी है।
पुस्तकालय सबके लिए सुलभ हों, शुल्क कम हों, ताकि साधारण व्यक्ति भी उसका लाभ उठा सके। उसमें अच्छी पुस्तकों का संग्रह हो। पुस्तकालय दिनभर खुला हो। सचमुच पुस्तकालय ज्ञान का दीपक है। भारत के प्रत्येक गाँव में पुस्तकालय होने चाहिए।
राष्ट्रीय एकता।
आज हमारा भारत देश स्वतंत्र है। इस स्वतंत्रता को पाने के लिए हम सभी भारतवासियों ने एकता के सूत्र में बंधकर ही प्रयास किया था। क्योंकि एकता में बहुत बड़ी ताकत होती है। अलग-अलग पाँच लकड़ियों को मिनटों में तोड़ा जा सकता है, परन्तु उन्हीं पाँच लकड़ियों के एक गढे को तोड़ना नामुमकिन है। यह है एकता की शक्ति।
व्यक्ति-व्यक्ति में एकता, समाज-समाज में एकता जैसे होती है, वैसे ही ‘राष्ट्रीय-एकता’ की बहुत आवश्यकता है। बिना एकता के कोई भी कार्य सफल नहीं हो सकता। बिना एकता के कोई संस्था अथवा संगठन भी खड़ा नहीं हो सकता। जिस देश में एकता नहीं होती है, वह कदापि विकास नहीं कर सकता, वह कदापि राष्ट्रोन्नति में सफल नहीं हो सकता।
हमारे देश में करीब पाँच सौ वर्षों तक मुगलों ने और करीब दो सौ वर्षों तक अंग्रेजों ने राज्य किया। इसका कारण भी यही है कि हममें (भारतीयों में) उस समय एकता नहीं थी। उस अनेकता के कारण ही हम गुलाम रहे और हमने बहुत-कुछ खोया।
जब हम जागृत हुए, हमारा सोया हुआ भारत जाग उठा और हमने एकता का महामंत्र फूंका तो नतीजा यह हुआ कि शत्रु भाग खड़े हुए। एकता से आजादी मिली, एकता से हमने विकास किया, एकता से हम आगे बढे और एकता से ही हम फिर विश्वगुरु बनने के लिए अग्रसर हैं। संगठन में शक्ति होती है, यह समझकर ही हम ‘राष्ट्रीय एकता’ के प्रहरी बने हुए हैं।
कम्प्यू टर
आज का युग कंप्यूटर का युग है। कंप्यूटर एक ऐसा मशीनी मस्तिष्क है जो ऐसे-ऐसे कार्य करने में सक्षम हैं जो मनुष्य की पहुंच के बाहर हैं। लाखों, करोड़ों व इससे भी अधिक संख्याओं की गणनाएँ यह पलक झपकते ही करने की क्षमता रखता है।
सबसे बड़ी विशेषता इसकी यह है कि भले ही मनुष्य इसके उपयोग में किसी प्रकार की कोई गलती कर दे, लेकिन यह सही सूचना देने में पूर्णतया सफल होता है।
आज कंप्यूटर सभी कार्यालयों, विभागों, विद्यालयों, बैंक, रेल्वे स्टेशन, बस-स्टैंड, हवाई अड्डों, दुकानों तक में पूर्ण सहयोगी है। मनोरंजन के साधनों में भी इसका जवाब नहीं। बच्चों के अलावा बड़े भी इसके मनोरंजन से प्रसन्न रहते हैं।
किसी भी व्यक्ति को यदि बंद कमरे में कंप्यूटर देकर बिठा दिया जाए तो शायद वह स्वतंत्र घूमने वाले प्राणी से अधिक प्रसन्न होगा। वैज्ञानिक क्षेत्र में देश-विदेश व अंतरिक्ष के कार्य कंप्यूटर के सहारे ही होते हैं। अंतरिक्ष के संबंध भी यही स्थापित करता है। इंटरनेट इसकी विशेष देन है। कुछ ही क्षणों में हम अपने प्रत्येक कार्य हेतु पूरे विश्व से सूचनाएँ प्राप्त कर सकते हैं। चिकित्सा के क्षेत्र में हर बीमारी की जाँच कंप्यूटर बखूबी करता है। लेकिन कई बार बच्चे व कुछ लोग इसका गलत प्रयोग कर स्वयं ही गुमराह होते हैं, जोकि गलत है। हमें इस उपयोगी साधन का सकारात्मक रूप से ही प्रयोग करना चाहिए।
खेलों का महत्व।
जीवन में खेलों का महत्वपूर्ण स्थान है। स्वास्थ्य के लिए खेल जरूरी है। मानसिक शांति के लिए भी खेल जरूरी है। खेल कई तरह के होते हैं। जैसे – देशी खेल और विदेशी खेल। आंतरिक खेल और बाहरी (मैदानी) खेल।
प्रायः देखा जाता है कि गाँवों में गुल्ली-डंडा, लंगड़ी, आँख-मिचौनी, चील-झपट, खो-खो, कबड्डी जैसे कई खेल खेले जाते हैं। इन खेलों से मनोरंजन तो होता ही है, साथ-साथ इनसे हमारा शरीर स्वस्थ, हृष्ट-पुष्ट भी रहता है। गाँवों में और भी कई मजेदार खेल खेले जाते हैं। आजकल गाँवों के खेल शहरों में खेले जाने लगे हैं। इससे प्रेम, भाईचारा बढ़ता है।
देशी खेलों के अतिरिक्त विदेशी खेल भी खेले जाते हैं। जैसे – क्रिकेट, हॉकी, फुटबॉल, वॉलीबाल आदि। विदेशी खेल देशी खेलों से महँगे होते हैं। खेलों को बढ़ावा देने के लिए स्कूल-कॉलेजों में प्रतियोगिताएँ, टूर्नामेंट आदि होते हैं। जीतनेवाले उत्तम खिलाड़ियों को प्रोत्साहित करने के लिए उन्हें पुरस्कार दिए जाते हैं, उनका सम्मान किया जाता है। अनेक बड़ी-बड़ी कंपनियाँ अच्छे खिलाड़ियों को अपने यहाँ नौकरी भी देती हैं।
खेल में खेल की भावना हो तो उसका कुछ मजा ही अलग है। खेल भी शिक्षा का आवश्यक अंग माना गया है। इससे खेलों का महत्व बढ़ गया है।
समय का सदुपयोग।
समय एक अनमोल वस्तु है। खोया हुआ समय कभी हाथ नहीं आता। हमारे जीवन की सफलता का आधार समय के सदुपयोग पर है। जो व्यक्ति समय का सदुपयोग करता है, वह अपने जीवन में उज्ज्वल सिद्धि प्राप्त कर सकता है। . जो लोग समय के महत्व को नहीं समझते हैं, वे अपने समय का दुरुपयोग करते हैं। सुबह देरी से उठना और उठने के बाद आधा घंटा आलस्य मिटाने में लगाना, गप्पें हाँकना, दूसरों की निंदा करना आदि में अपना काफी समय बरबाद करना ये सब उचित नहीं।
यदि हम महापुरुषों की जीवनियाँ पढ़ेंगे, तो समय के महत्व का पता चलेगा। समय के सदुपयोग ने ही उनको अमर कीर्ति प्रदान की है। ज्ञान, धन, कीर्ति, कुशलता आदि प्राप्त करने के लिए समय का सदुपयोग करना अनिवार्य है।
समय का सदुपयोग करने के लिए हमें प्रत्येक काम ठीक समय पर करना चाहिए। अध्ययन, व्यायाम, समाजसेवा, मनोरंजन और अन्य सभी काम ठीक-ठीक समय पर करना चाहिए। ऐसा करने पर काम का बोझ महसूस नहीं होगा। समय को दुर्लभ सम्पत्ति मानकर उसका सदुपयोग करना चाहिए।
अनुशासन की आवश्यकता।
अनुशासन का अर्थ है – शासन का अनुसरण करना अर्थात् शासन के नियम और नियंत्रण के अधीन रहना। अनुशासित जीवन ही सच्चा जीवन माना जाता है। जीवन के प्रत्येक क्षेत्र में अनुशासन का होना नितांत जरूरी है।
यदि हम अनुशासन का पालन नहीं करेंगे, नियमों को तोड़ेंगे, तो फिर गड़बड़ी हो जायेगी और चारों तरफ अव्यवस्था फैल जायेगी। अतः व्यवस्था को बनाये रखने के लिए अनुशासन का होना जरूरी है।
अनुशासन किसी एक क्षेत्र के लिए सीमित नहीं है। अनुशासन नीचे से ऊपर तक, छोटे से बड़े तक, अमीर से गरीब तक, प्रजा से शासक तक अर्थात् प्रत्येक क्षेत्र में यत्र-तत्र-सर्वत्र अनुशासन की आवश्यकता है। यदि विद्यार्थी को विद्यालय में, खेल के मैदान में, छात्रावास में, घर-परिवार में, सार्वजनिक स्थानों में पालन करने का अभ्यास हो जाए तो वह जीवन भर अनुशासित रहेगा। अतः जरूरी है कि अध्यापक विद्यार्थियों को अनुशासन सिखाएँ।
कहते हैं, बाल्यावस्था में जो अच्छी बातें सीख ली जाती हैं, उनका अच्छा असर भी जीवन-पर्यंत होता है। अतः अच्छे नागरिक बनने के लिए भी अनुशासन की आवश्यकता है और अच्छे शासक के लिए भी अनुशासन की आवश्यकता है।
नागरिक के कर्तव्य।
प्रत्येक नागरिक अपने जीने के लिए सदा कुछ अधिकारों की इच्छा रखता है। उसे यह भी जानना होगा कि अधिकारों की माँग के साथ-साथ कुछ कर्तव्यों का भी पालन करना पड़ता है। यह बात एक परिवार, एक समाज, एक गाँव या शहर तथा एक देश के लिए भी लागू होती है।
भारत 15 अगस्त 1947 को स्वतंत्र हुआ। हम स्वतंत्र देश के नागरिक हैं। जब हम गुलाम थे, तो न हमें कोई अधिकार था और न कोई कर्तव्य । अब हम स्वतंत्र देश के स्वतंत्र नागरिक होने के नाते हमें कुछ मौलिक अधिकार मिले हैं, तो कुछ मौलिक कर्तव्य भी।
अधिकार और कर्तव्य एक सिक्के के दो पहलू के समान हैं। बिना कर्तव्य के अधिकार का कोई महत्व नहीं है। कर्तव्य अधिकार का एक अभिन्न अंग है। जो किसी एक व्यक्ति के लिए कर्तव्य है, वही दूसरे के लिए अधिकार है।
भारतीय संविधान के भाग-4क, अनुच्छेद 51 क में मूल कर्तव्य शामिल किए गए हैं। इसके अनुसार भारत के प्रत्येक नागरिक का यह कर्तव्य होगा कि वह –
उपर्युक्त नागरिक कर्तव्यों का पालन कराने या उनका उल्लंघन होने पर दंड देने के लिए संविधान में कोई उपबंध नहीं है। मूल कर्तव्यों का पालन कराने का एकमात्र तरीका यह है कि लोगों को नागरिकता के मूल्यों तथा कर्तव्यों के बारे में शिक्षित किया जाए और उनमें पर्याप्त जागृति उत्पन्न की जाए तथा एक ऐसे अनुकूल वातावरण का निर्माण किया जाए जिसमें प्रत्येक नागरिक अपने सवैधानिक कर्तव्यों का पालन करने तथा समाज के प्रति अपना ऋण चुकाने में गर्व तथा बंधन का अनुभव करे।
इंटरनेट – वरदान है या अभिशाप / इंटरनेट से लाभ / इंटरनेट का महत्व।
अर्थ : इंटरनेट विश्व के विभिन्न स्थानों पर स्थापित कम्प्यूटरों के कई अंतर्जालों का एक दूसरे से संबंध स्थापित करने का जाल है, जिस पर सूचना एक स्थान से दूसरे स्थान तक शीघ्र पहुँच जाती है। इंटरनेट के द्वारा सूचनातंत्र मानव की मुट्ठी में बंद होता जा रहा है।
लाभ : इंटरनेट ने मीडिया और समाचार माध्यमों के पूरे कार्य पर व्यापक प्रभाव डाला है। इंटरनेट ने हर प्रकार के व्यवसाय को एक नया स्वरूप प्रदान किया है। विश्व के अधिकांश समाचार पत्र ऑनलाइन हो गये है। अनेक ऐसी वेबसाइट हैं, जो ऑनलाइन शिक्षा, मनोरंजन आदि प्रदान कर रही हैं।
दुष्परिणामः इंटरनेट ने जहाँ युवा पीढ़ी को फायदा पहुंचाया है तो इसके नुकसान भी है। विद्यार्थी अपना अधिकांश समय व्हाट्सएप्प, फेसबुक, ट्विटर पर बिताते हैं। इंटरनेट ने मनुष्य के जीवन में गोपनीयता को भी खत्म किया है। इंटरनेट पर अनेक अश्लील साहित्य मौजूद है जिससे आज की युवा पीढ़ी पर बुरा असर पढ़ रहा है। कई बार इंटरनेट के माध्यम से जनता को भ्रमित भी कर दिया जाता है।
उपसंहार : इंटरनेट ने आज पूरी दुनियाँ को एक विश्वग्राम (Global Village) में बदल दिया है। इंटरनेट के माध्यम से सूचना की उपलब्धता बहुत आसान हो गयी है। हमें इंटरनेट के सही उपयोग पर ध्यान देना चाहिए।
स्वच्छ भारत अभियान / स्वच्छता का महत्व।
स्वस्थ जीवन जीने के लिए स्वच्छता का विशेष महत्व है। आरोग्य को नष्ट करने के जितने भी कारण हैं, उनमें गन्दगी प्रमुख है। बीमारियाँ गन्दगी में ही पलती हैं। जहाँ कूड़े-कचरे के ढेर जमा रहते हैं, मल-मूत्र सड़ता है, नालियों में कीचड़ भरी रहती है, सीलन और साड़न बनी रहती है, वहीं मक्खी, पिस्सू, खटमल जैसे बीमारियाँ उत्पन्न करने वाले कीड़े उत्पन्न होते हैं। कहना न होगा कि हैजा, मलेरिया, दस्त, पेट के कीड़े, चेचक, खुजली, रक्त-विकार जैसे कितने ही रोग इन मक्खी, मच्छर जैसे कीड़ों से ही फैलते हैं।
महात्मा गांधी ने अपने आसपास के लोगों को स्वच्छता बनाए रखने संबंधी शिक्षा प्रदान कर राष्ट्र को एक उत्कृष्ट संदेश दिया था। उन्होंने ‘स्वच्छ भारत’ का सपना देखा था। वे चाहते थे कि भारत के सभी नागरिक एक साथ मिलकर देश को स्वच्छ बनाने के लिए कार्य करें। महात्मा गांधी के स्वच्छ भारत के स्वप्न को पूरा करने के लिए, प्रधानमंत्री श्री नरेन्द्र मोदी जी ने 2 अक्टूबर 2014 को स्वच्छ भारत अभियान शुरू किया है। इस अभियान का उद्देश्य अगले पांच वर्ष में स्वच्छ भारत का लक्ष्य प्राप्त करना है ताकि बापू की 150 वीं जयंती को इस लक्ष्य की प्राप्ति के रूप में मनाया जा सके।
शहरी क्षेत्रों के लिए स्वच्छ भारत मिशन का उद्देश्य 1.04 करोड़ परिवारों को लक्षित करते हुए 2.5 लाख समुदायिक शौचालय, 2.6 लाख सार्वजनिक शौचालय, और प्रत्येक शहर में एक ठोस अपशिष्ट प्रबंधन की सुविधा प्रदान करना है। ग्रामीण क्षेत्रों के लिए स्वच्छ भारत मिशन का उद्देश्य पांच वर्षों में भारत को खुला शौच से मुक्त बनाना है। अभियान के तहत देश में लगभग 11 करोड़ 11 लाख शौचालयों के निर्माण के लिए एक लाख चौंतीस हजार करोड़ रुपए खर्च किये जाएंगे।
स्वच्छ भारत अभियान का उद्देश्य केवल आसपास की सफाई करना ही नहीं है अपितु नागरिकों की सहभागिता से अधिक-से-अधिक पेड़ लगाना, कचरा मुक्त वातावरण बनाना, शौचालय की सुविधा उपलब्ध कराकर एक स्वच्छ भारत का निर्माण करना है। देश में पर्यटन को बढ़ावा देने के लिए स्वच्छ भारत का निर्माण करना अत्यंत महत्त्वपूर्ण है।
सरकार जन समुदाय की सक्रिय भागीदारी के बिना किसी भी अभियान में सफलता प्राप्त नहीं कर सकती। इसके लिए व्यापक जन चेतन कार्यक्रम चलाना होगा। स्कूली विद्यार्थीयों को अपने घर तथा पड़ोस के सभी घरों में शौचालय का होना तथा इसका उपयोग सुनिश्चित करना चाहिए। उनको अपने घर तथा आस पड़ोस में अच्छी आदतों को फैलाना चाहिए। घर, गली, स्कूल तथा अन्य सभी सार्वजनिक स्थानों पर गन्दगी नहीं फैलाना चाहिए तथा कचरा पात्र में ही कचरा डालना चाहिए। खुले में शौच नहीं जाना चाहिए।
अस्वच्छ भारत की तस्वीरें भारतीयों के लिए अक्सर शर्मिंदगी की वजह बन जाती है। इसलिए स्वच्छ भारत के निर्माण एवं देश की छवि सुधारने का यह समय और अवसर है। यह अभियान न केवल नागरिकों को स्वच्छता संबंधी आढ़तें अपनाने बल्कि हमारे देश की छवि स्वच्छता के लिए तत्परता से काम कर रहे देश के रूप में बनाने में भी मदद करेगा।
मेरा प्रिय कवि/साहित्यकार।
मेरा प्रिय कवि है रवीन्द्रनाथ ठाकुर। रवीन्द्रनाथ ठाकुर केवल साहित्यकार ही नहीं शिक्षणतज्ञ भी थे। उन्होंने साहित्य में ही नहीं शिक्षा के क्षेत्र में भी अनुपम योगदान दी है।
रवीन्द्रनाथ ठाकुर का जन्म 7 मई 1861 के दिन बंगाल प्रांत में हुआ था। इनके पिता महर्षि देवेन्द्रनाथ ब्रह्मसमाज के बड़े नेता थे। रवीन्द्र ने प्रारंभिक शिक्षा कॉन्वेंट स्कूल में पायी थी। बाद में उन्होंने घर पर ही शिक्षा प्राप्त की। सत्रह वर्ष की आयु में पढ़ाई करने के उद्देश्य से इंग्लैंड गये और वहाँ यूनिवर्सिटी आफ लंदन में दाखिला लिया।
बीसवीं सदी के आरंभ में रवीन्द्रनाथ बंगाल के राष्ट्रीय आंदोलन के नेता थे। किंतु उन्हें राजनीति पसंद नहीं आयी। वे तो जन्मजात साहित्यकार तथा कवि थे। उन्होने अत्यधिक निबंध, नाटक, कविताएँ, कहानियाँ तथा उपन्यास लिखे। उनके साहित्य में राजनीति, शिक्षा, धर्म, कला आदि विषयों के मौलिक लेख है। उनका बंगाली साहित्य में भी अत्यधिक योगदान रहा। रवीन्द्र श्रेष्ठ चित्रकार भी थे। सन 1908 में वे बंगला साहित्य सम्मेलन के सभापति चुने गये।
1912 में पुनः इंग्लैण्ड गए तथा ‘गीतांजली’ का अंग्रेजी में अनुवाद किया। सन् 1913 में इस रचना के लिए उन्हे नोबेल पुरस्कार दिया गया। उनकी रचनाएँ है – ‘गीतांजली’, ‘नैवेद्य’ (काव्य), ‘काबुलीवाला’, ‘सुभा’, ‘क्षुधित पाषाण’ (कहानी संग्रह), ‘डाकघर’, ‘राजा’ (नाटक) इत्यादि । रवीन्द्रनाथ ठाकुर सर्वोच्च नोबेल पुरस्कार से अलंकृत है। 1913 वर्ष में ही उन्हें ‘सर’ की उपाधि दी गई। रवीन्द्रनाथ ठाकुर का बंगला साहित्य के साथ-साथ अंग्रेजी साहित्य में भी उच्च स्थान रहा है। उनकी कविताओं का अनुवाद संसार के अनेक भाषाओं में हो चुका है।
स्वास्थ्य और व्यायाम।
मानसिक सुख को प्राप्त करने का मुख्य साधन शारीरिक स्वास्थ्य है। मानव जीवन में स्वास्थ्य ही सर्वस्व हैं। शरीर को स्वस्थ रखने के लिए लोग अनेक उपाय करते हैं पर उनमें सबसे अधिक सरल और सुगम उपाय व्यायाम है। जीवन व्यायाम के बिना कभी स्फूर्तिमय नहीं रहता। इसलिए व्यायाम मानव जीवन के लिए अत्यधिक आवश्यक है।
शारीरिक अंगों द्वारा समुचित ढंग से परिश्रम करने को व्यायाम कहते हैं। व्यायाम के विभिन्न भेद हैं। भिन्न-भिन्न प्रकार के आसन, दण्ड-बैठक, खुले मैदान में दौड़ना, घूमना, प्राणायाम करना, कुश्ती लड़ना, तैरना, हॉकी, फुटबाल, वालीबाल, क्रिकेट, टेनिस, कबड्डी, बेडमिन्टन खेलना आदि सभी को व्यायाम के अन्तर्गत रख सकते हैं। इन व्यायामों में शरीर के विभिन्न अंगों से समुचित काम लिया जाता है जिससे मांस-पेशियों में बल आता है और उनका विकास समुचित तरीके से होता है। हड्डियों में मजबूती आती है तथा परिश्रम करने से पसीना अधिक निकलता है जिससे रक्त का संचार ठीक ढंग से होता है और वह साफ हो जाता है।
मानव-जीवन में व्यायाम का विशेष स्थान है। जिस प्रकार रेल के इंजन को चलाने के लिए कोयले और पानी की आवश्यकता होती है, उसी प्रकार शरीर को क्रियाशील बनाये रखने के लिए व्यायाम रूपी कोयले की आवश्यकता होती है। व्यायाम से सारा शरीर सुडौल, सुगठित एवं दृढ़ बन जाता है। रक्त संचार ठीक तरह तथा तीव्र गति से होता है। हृदय की गति में वेग पैदा हो जाता है तथा पाचक शक्ति भी अपना कार्य ठीक तरह से करती है। सभी इन्द्रियाँ ठीक तरह से अपना कार्य करती रहती हैं। हृदय में उत्साह, आत्म-विश्वास तथा निडरता रहती है। मन भी कभी अप्रसन्न नहीं होता। रोग तो व्यायामशील व्यक्ति के पास फटक ही नहीं सकते।
व्यायाम का सबसे अधिक प्रभाव व्यक्ति के मस्तिष्क पर पड़ता है। व्यायाम द्वारा मस्तिष्क का विकास होता है। अंग्रेजी में कहावत है – ‘Healthy mind in a healthy body’ अर्थात् स्वस्थ शरीर में स्वस्थ मस्तिष्क होता है। व्यायाम द्वारा शरीर में एक अनुपम स्फूर्ति का श्रोत बहने लगता है।
आज के वैज्ञानिक युग में व्यायाम अत्यंत आवश्यक है। व्यायाम के लिए खुली हवा और खुली जगह आवश्यक होती है ताकि श्वास लेने के लिए स्वच्छ वायु मिल सके। शरीर के लिए व्यायाम अत्यंत आवश्यक है। जीवन में अधिक समय तक सुखी और निरोग रहने का एक मात्र साधन व्यायाम ही है।
बेटी बचाओ, बेटी पढ़ाओ।
हमें हमारे देश और हमारी संस्कृति पर हमेशा गर्व रहा है। हजारों वर्षों से हमारे देश ने किसी दूसरे देश पर आक्रमण नहीं किया। हम शांतिप्रिय रहे हैं। ऐसी महान सांस्कृतिक विरासत होने के बावजूद हमारे समाज में एक ऐसी बुराई है जो आज पूरे संसार के सामने हमें हमारी नजरें नीची करने के लिए मजबूर कर देती हैं। वो बुराई है पुरुषों की तुलना में स्त्री को दोयम दर्जे का स्थान देना। हमारा समाज इतना ज्यादा पुरुषप्रधान हो गया है कि आज देश की जनसंख्या का बड़ा हिस्सा बेटी पैदा ही नहीं करना चाहता। इसीका नतीजा है कि हमारे देश में पुरुषों के मुकाबले स्त्रियों की संख्या घटती जा रही है। इसी वजह से हमारे प्रधानमंत्री श्री नरेंद्र मोदी जी ने ‘बेटी बचाओ, बेटी पढ़ाओ’ योजना की शुरुआत की।
‘बेटी बचाओ, बेटी पढ़ाओ’ योजना को एक राष्ट्रीय अभियान के माध्यम से कार्यान्वित किया गया है। इसका मुख्य उद्देश्य है कन्या भ्रूण हत्या की रोकथाम, बालिकाओं के अस्तित्व को बचाना, उनकी सुरक्षा सुनिश्चित करना तथा बालिकाओं की शिक्षा और भागीदारी सुनिश्चित करना। यह योजना न केवल लड़कियाँ बल्कि पूरे समाज के लिए एक वरदान साबित हो सकती है।
वर्तमान समय में अजन्मे बच्चे के लिंग का पता लगाने की सुविधा आसानी से उपलब्ध है। इस वजह से कन्या भ्रूण हत्या के मामलों में तेजी से वृद्धि हुई है। लोग सोचते हैं कि लड़का बड़ा होकर पैसा कमाएगा। लड़की इसके विपरीत, दहेज़ लेकर घर से जाएगी। इस तरह के आर्थिक कारणों से लड़कियों के विरुद्ध सामाजिक पक्षपात होता रहा है। समाज में गहरे तक यह बात बैठी हुई है कि लड़कियाँ पैदा होते ही बड़ी जिम्मेदारी गले आ जाती है। इन कारणों से लिंगानुपात को नुकसान पहुंचा है। महिलाओं के जन्म से पहले ही उनके अधिकारों का हनन शुरू हो जाता है तथा जन्म के बाद भी उनके साथ भेदभाव नहीं थमता।
स्वास्थ्य, पोषण और शिक्षा की जरूरतों को लेकर उनके साथ कई तरह से पक्षपात होता है। लड़कियाँ को बोझ की तरह देखा जाता है जबकि वास्तविकता इसके विपरीत है। महिला सशक्तिकरण से समाज को पिछड़ेपन से मुक्ति मिलती है। यह ठीक ही कहा गया है कि एक पुरुष को शिक्षा देने से वह अकेला शिक्षित होता है लेकिन स्त्री की शिक्षा से समूचा परिवार अज्ञान के अंधेरे से निकल कर शिक्षित हो सकता है। एक शिक्षित माँ अपने बच्चों को शिक्षा और संस्कार तो देती ही है, उनके स्वास्थ्य का भी बेहतर ध्यान रखती है।
शिक्षित स्त्री घर-परिवार का प्रबंधन अधिक कुशलतापूर्वक कर सकती है, आय-अर्जन तथा अन्य कामों के मामले में पति का हाथ बँटाती है, अपने अधिकारों और दायित्वों को बेहतर रूप से समझती है और समाज में फैली कई बुराइयों के विरोध में खड़ी हो सकती है। इसलिए आज के समय में यह जरूरी है कि लड़कियों को लेकर शहरी तथा ग्रामीण भागों के लोगों के बीच फैली अंधविश्वासी मान्यताओं और प्रथाओं को खत्म किया जाए। कहीं पर भी कन्या भ्रूण हत्या हो रही हो तो तुरंत इसकी जानकारी पुलिस को दें। लोगों को इस बारे में सजग करे।
इसके लिए मीडिया और संचार के नए तरीकों का पूरी तरह से इस्तेमाल करने की आवश्यकता है। ‘बेटी बचाओ, बेटी पढ़ाओ’ अभियान इसी लक्ष्य को हासिल करने, इसके बारे में जागरूकता फैलाने और लोगों की मानसिकता में बदलाव लाने के लिए शुरू किया गया है।
‘बेटी बचाओ, बेटी पढ़ाओ’ सिर्फ एक योजना नहीं बल्कि देश के हर नागरिक की सामूहिक जिम्मेदारी है। यदि एक समाज के रूप में हम इस समस्या के प्रति संवेदनशील नहीं होंगे, जागरूक नहीं होंगे, तो हम अपनी ही नहीं, आने वाली पीढ़ी के लिए भी एक भयंकर संकट को निमंत्रण देंगे।
पर्यावरण की रक्षा।
प्रस्तावना : पर्यावरण की रक्षा से तात्पर्य पर्यावरण की सुरक्षा करना है। वृक्ष, वनस्पतियों का मनुष्य के जीवन में अत्यधिक महत्व है। एक शांतिपूर्ण और स्वस्थ जीवन जीने के लिए एक स्वच्छ वातावरण बहुत ही जरूरी है। पर्यावरण के बिना हम जीवन की कल्पना भी नहीं कर सकते। आज पर्यावरण मनुष्य की लापरवाही के कारण खराब हो रहा है। परिणामतः अनेकों समस्याएँ मनुष्य के सामने उपस्थित हो रही है। अगर पर्यावरण का प्रदूषण बढ़ता गया तो इस धरती पर मानव जाति के साथ-साथ समस्त जीवों के लिए भी खतरा उत्पन्न हो जाएगा।
रक्षा के उपायः पर्यावरण को बेहतर बनाने के लिए हमें सबसे पहले ‘जल’ को प्रदूषण से बचाना होगा। कारखानों का गंदा पानी, सीवर लाइन का गंदा जल नदियों और समुद्र में गिरने से रोकना होगा।
जल प्रदूषण की तरह वायु को भी प्रदूषित होने से रोकना होगा। कारखानों से, वाहनों से निकलने वाले धुएं से बचने के लिए दूसरे रास्तें खोजने होंगे। विद्युत से चलने वाले वाहनों को हमें और बढ़ाना होगा। इसी तरह ध्वनि प्रदूषण भी विकराल समस्या है। हमें लोगों को इस संबंध में जागरूक करने की जरूरत है।
पहाड़ों एवं जंगलों की सुरक्षा पर ध्यान देने की जरूरत है। जंगल को कटने से बचाना होगा। खाली, बंजर जमीन पर पौधारोपण को बढ़ावा देना होगा।
उपसंहार : पर्यावरण की रक्षा जीवन की रक्षा है। इस धरती पर जीवन इसिलिए है क्योंकि यहाँ पर्यावरण है। आज पर्यावरण प्रदूषण दुनिया भर के लिए गंभीर मुद्दा है। हमें सक्रिय रूप से पर्यावरण की रक्षा में योगदान देना चाहिए। हमें अपने प्राकृतिक संसाधनों का उचित इस्तेमाल करना चाहिए। उपभोक्तावादी संस्कृति एवं प्रकृति के अंधाधुंध उपयोग से बचना होगा।
मेरा कर्नाटक महान / कर्नाटक का वैभव।
कर्नाटक भारत का आठवां सबसे बड़ा राज्य है। इसकी जनसंख्या करीब छः करोड़ है। कर्नाटक की प्राकृतिक सुषमा नयन मनोहर है। कर्नाटक राज्य में तीन प्रधान मंडल हैं: तटीय क्षेत्र करावली, पहाड़ी क्षेत्र मलेनाडु जिसमें पश्चिमी घाट आते हैं, तथा तीसरा बयलुसीमी क्षेत्र जहां दक्खिन पठार का क्षेत्र है। राज्य का अधिकांश क्षेत्र बयलुसीमी में आता है और इसका उत्तरी क्षेत्र भारत का सबसे बड़ा शुष्क क्षेत्र हैं।
कर्नाटक की आधिकारिक और सर्वाधिक बोली जाने वाली भाषा कन्नड़ है और इसकी राजधानी बेंगलूरु है। बेंगलूरु शिक्षा, उद्योग तथा विज्ञान का केन्द्र माना जाता है। इसे ‘सिलिकॉन सिटी’ भी कहते हैं। यह भारत में हो रही त्वरित आर्थिक एवं प्रौद्योगिकी का अग्रणी योगदानकर्ता है। भारत में कर्नाटक जैवप्रौद्योगिकी के क्षेत्र में भी अग्रणी है।
सर सी.वी. रामन, सर एम. विश्वेश्वरय्या, डॉ. सी.एन.आर. राव, डॉ. शकुंतला देवी जैसे दिग्गजों ने वैज्ञानिक तथा प्रौद्योगिकी के क्षेत्र में नारायण मूर्ति ने कर्नाटक का नाम रोशन किया है। सन् 2013 में डॉ. सी.एन.आर. राव को ‘भारत रत्न’ से सम्मानित किया गया है।
कर्नाटक में सोना, ताँबा, लोहा आदि कई प्रकार की उपयोगी धातुएँ मिलती हैं। कागज, लोहे. और इस्पात के कारखाने हैं। चीनी, सिमेंट, रेशम के भी कारखाने हैं। कर्नाटक को ‘चंदन का आगार’ भी कहते हैं। भारत के अग्रणी बैंकों में से सात बैंकों का उद्गम इसी राज्य से हुआ था। भारत के. करोड़ों रेशम उद्योग से अधिकांश भाग कर्नाटक राज्य में आधारित है। यहाँ की बेंगलोर सिल्क और मैसूर सिल्क विश्वप्रसिद्ध हैं।
अपने विस्तृत भूगोल, प्राकृतिक सौन्दर्य एवं लम्बे इतिहास के कारण कर्नाटक राज्य बड़ी संख्या में पर्यटन आकर्षणों से परिपूर्ण है। राज्य के मैसूर में स्थित महागाजा पैले” इतना आलीशान एवं खूबसूरत बना है, कि उसे विश्व के दस कुछ सुंदर महलों में गिना जाता है। कर्नाटक के पश्चिमी घाट में आनेवाले तथा दक्षिणी जिलों में प्रसिद्ध पारिस्थितिकी पर्यटन स्थल हैं जिनमें कुद्रेमुख, मडिकेरी तथा आगुम्बे आते हैं। राज्य में 25 वन्य जीवन अभयारण्य एवं 5 राष्ट्रीय उद्यान हैं। इनमें से कुछ प्रसिद्ध हैं बंडीपुर राष्ट्रीय उद्यान, बनेरघट्टा राष्ट्रीय उद्यान एवं नागरहोले राष्ट्रीय उद्यान।
हम्पी में विजयनगर साम्राज्य के अवशेष तथा पत्तदकल में प्राचीन पुरातात्विक अवशेष युनेस्को विश्व धरोहर चुने जा चुके हैं। इनके साथ ही बादामी के गुफा मंदिर तथा ऐहोले के पाषाण मंदिर बादामी चालुक्य स्थापत्य के अद्भुत नमूने हैं तथा प्रमुख पर्यटक आकर्षण बने हुए हैं। यहाँ बने गोलगुम्बज तथा इब्राहिम रौजा दक्खन सल्तनत स्थापत्य शैली के अद्भुत उदाहरण हैं। श्रवणबेलगोला में स्थित गोमटेश्वर की 18 मीटर ऊंची मूर्ति विश्व की सर्वोच्च एकाश्म प्रतिमा है। हाल के कुछ वर्षों में कर्नाटक स्वास्थ्य रक्षा पर्यटन हेतु एक सक्रिय केन्द्र के रूप में भी उभरा है। राज्य में देश के सर्वाधिक स्वीकृत स्वास्थ्य प्रणालियाँ और वैकल्पिक चिकित्सा उपलब्ध हैं।
कर्नाटक का विश्वस्तरीय शास्त्रीय संगीत में विशिष्ट स्थान है, जहां संगीत की कर्नाटिक और हिन्दुस्तानी शैलियाँ स्थान पाती है। राज्य में दोनों ही शैलियाँ के पारंगत कलाकार हुए हैं। वैसे कर्नाटिक संगीत में कर्नाटिक नाम कर्नाटक राज्य विशेष का ही नहीं, बल्कि दक्षिण भारतीय शास्त्रीय संगीत को दिया गया है।
कर्नाटिक संगीत के कई प्रसिद्ध कलाकार जैसे गंगूबाई हंगल, मल्लिकार्जुन मंसूर, भीमसेन जोशी, बसवराज राजगुरु, सवाई गंधर्व और कई अन्य कर्नाटक राज्य से हैं और इनमें से कुछ को कालिदास सम्मान, पद्म भूषण और पद्म विभूषण से भी भारत सरकार ने सम्मानित किया हुआ है।
राज्य में भारत के कुछ प्रतिष्ठित शैक्षिक और अनुसंधान संस्थान भी स्थित हैं, जैसे भारतीय विज्ञान संस्थान, भारतीय प्रबंधन संस्थान, राष्ट्रीय प्रौद्योगिकी संस्थान, आई.आई.टी., कर्नाटक और भारतीय राष्ट्रीय विधि विश्वविद्यालय आदी।
बसवण्णा, अक्कमहादेवी, अल्लमप्रभु, सर्वज्ञ जैसे संतों ने अपने अनमोल वचनों से मार्गदर्शन किया है, तो पुरंदरदास, कनकदास आदि भक्त कवियों ने नीति और भक्ति की सरिता का प्रवाह किया है। इतना ही नहीं, पंपा, रन्ना, पोन्ना, कुमारव्यास, हरिहर, राघवांक जैसे कवियों ने कन्नड़ साहित्य को समृद्ध किया है।
वर्तमान में कुवेंपु, बेंद्रे, शिवराम कारंत, मास्ति वेंकटेश अय्यंगार, गोकाक, अनन्तमूर्ति, कार्नाड तथा कंबार दिग्गज साहित्यकारों को ज्ञानपीठ पुरस्कार प्राप्त हुआ है। यह हमारे कर्नाटक राज्य के लिए गौरव का विषय है। इन्ही कारणों से मेरा कर्नाटक महान है और मुझे अपने कर्नाटक राज्य पर बहुत गर्व है।
ग्राम सुधार।
भारत की करीब 70 प्रतिशत जनसंख्या गाँवों में निवास करती है। गाँव का नाम आते ही हमारे मन में एक सुंदर-सी कल्पना जन्म लेने लगती है – अपूर्व शांति की भावना उत्पन्न करने वाले हरेभरे खेत, स्वच्छ वायुमंडल और नीरवता को चीरती हुई पक्षियों की मीठी-मीठी चहक। यही नहीं दही बिलौने और मट्ठा मथने के काम में लगी गाँव की औरतें तथा हल और बैल लेकर खेतों की ओर जाते हुए किसान की तस्वीर मन में उभर आती है। लेकिन आज का गाँव हमारी कल्पना के ठीक विपरीत है।
गाँव की गलियाँ बरसात में कीचड़ का अंबार उगलने लगती हैं। गाँव बीमारियों का घर बनते जा रहे हैं। भोले-भाले बच्चे अशिक्षित, अनपढ़ रह जाते हैं। इसके अतिरिक्त बनिए का ऋण उनकी सिर पर हमेशा सवार रहता है। आज हमें अपने गाँवों का आर्थिक और सांस्कृतिक दृष्टि से सुधार करना होगा। किसान को खेती करने के नए तरीकों से परिचित करवाना होगा। उन्हें साक्षर बनाने का प्रयास करना होगा। गावों में समाज सुधार के लिए संस्थाओं की स्थापना करनी होगी तथा सांस्कृतिक उन्नति के लिए ग्रामवासियों के स्वास्थ्य तथा शिक्षा पर ध्यान देना होगा।
सड़कों का निर्माण करना होगा, साथ ही साथ ग्रामीण उद्योग-धन्धों का विकास करना होगा। उनके जीवन को मनोरंजक बनाने के लिए अनेक कार्यक्रमों का आयोजन करना होगा। इन सभी प्रयासों के बाद ही गाँव वाले सुख का जीवन व्यतीत कर सकेंगे और तभी संपूर्ण देश उत्थान के शिखर पर प्रतिष्ठित होगा।
Students can Download Political Science Chapter 7 National Integration Questions and Answers, Notes, KSEEB Solutions for Class 9 Social Science helps you to revise complete Karnataka State Board Syllabus and score more marks in your examinations.
I. Fill in the blanks with suitable words:
Question 1.
India is a secular country and so does not oppose any ______
Answer:
religion.
Question 2.
Communalism is a major obstacle to ______
Answer:
“National Integration”.
![]()
Question 3.
Republic Day is a _____festival.
Answer:
National
Question 4.
India has considered _______ languages as National languages
Answer:
22
Question 5.
Our National Animal is ______ .
Answer:
Tiger
II. Answer the following questions after group discussion :
Question 1.
What is ‘Nationalism’?
Answer:
The peoples’ emotion of knowing their land as motherland, their brotherhood and their equal participation in all the times in the nation is ‘Nationalism’.
Question 2.
What do you mean by ‘National Integration’?
Answer:
The situation where all the people of the country feel they are one although there difference with regard to race, religion, culture and language is called “National Integration”.
Question 3.
Which are the factors that lead to unity in diversity?
Answer:
The important factors which enable unity in diversity are:
![]()
Question 4.
Which are the factors that promote National Integration?
Answer:
Many factors are there for promoting national integration. They are:
![]()
Question 5.
What are your suggestions to overcome obstacles that affect National Unity?
Answer:
The suggestions to overcome obstacles that affect National Unity are :