Students can Download Karnataka SSLC Science Model Question Paper 3 with Answers, Karnataka SSLC Science Model Question Papers with Answers helps you to revise the complete Karnataka State Board Syllabus and score more marks in your examinations.
Karnataka State Syllabus SSLC Science Model Question Paper 3 With Answers
Time: 3 Hours
Max Marks: 80
I. Four alternatives are provided for each of the following, questions or incomplete statements. Choose the most appropriate alternative and write with its alphabet. ( 8 × 1 = 8 )
Question 1.
In the following sketch of stomatal apparatus the parts I, II and III where labeled differently by four students, the correct labeling out of the following is
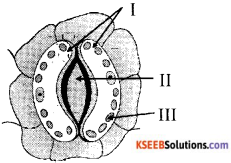
A) I Guard cell, II stomatal pore, III chloroplast.
B) I Stomatal pore, II guard cell, III chloroplast
C) I chloroplast, II stomatal pore, III Guard cells
D) I Guard cell, II chloroplast, III stomatal pores.
Answer:
A) I Guard cell, II stomatal pore, III chloroplast.
Question 2.
The sulphide ore among the following is
A) Haematite
B) Bauxite
C) Argentite
D) Zinc blende.
Answer:
D) Zinc blende.
Question 3.
Which of the following it most suitable for the core of electromagnets
A) Air
B) Soft iron
C) Steel
D) Cu-Ni alloy
Answer:
B) Soft iron
![]()
Question 4.
Decomposers of an ecosystem consists of
A) Organisms which make organic compound out of inorganic compounds
B) Organisms which use radiant energy to produce biomass
C) Certain blue green algae and algae
D) Certain fungi and bacteria.
Answer:
D) Certain fungi and bacteria.
Question 5.
Phototropism is caused to differential distribution of
A) Abscisic asid
B) Gibberellin
C) Cytokinin
D) Auxin
Answer:
D) Auxin
Question 6.
Most of the sources of energy we use represent stored solar energy which of the following is not ultimately derived from the sun’s energy
A) Geothermal energy
B) Wind energy
C) Nuclear energy
D) Bio mass
Answer:
C) Nuclear energy
Question 7.
Which of the following represent N2 molecules
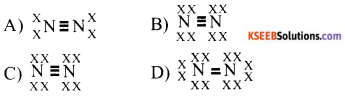
Answer:
![]()
![]()
Question 8.
Galvanisation of iron means coating iron with
A) Zinc
B) Nickle
C) Chromium
D) Tin
Answer:
A) Zinc
II. Answer the following questions ( 8 × 1 = 8 )
Question 9.
What is the principle of a electric motor ?
Answer:
An electric motor is based on the principle that when a current is passed through conductor placed a uniformed magnetic field it experiences a force which maker the conductor rotate.
Question 10.
Why does an aqueous solution of an acid conduct electricity ?
Answer:
The presence of hydrogen (H+) or hydronium (H3O+) ions in aqueous solution of an acid are responsible for conducting electricity.
Question 11.
Write a mathematical expression for Joule’s law of heating. Name on device which work on this principle.
Answer:
H = I2Rt
An electric heater works on Joule’s law of heating
![]()
Question 12.
How is the increase in demand for energy affecting our environment adversely ?
Answer:
The increase in demand for energy affects our environment adversely due to the increase pollutants like 2, CO, SO4 etc. are released into the atmosphere which lead to greenhouse affect.
Question 13.
If a person is working on a treadmill in a gymnasium will it effect his rate of breathing ? How ?
Answer:
Yes, it will affect his rate of breathing. It will become fast to supply more oxygen to meet the increased demand of energy.
Question 14.
When is the force experienced by current carrying conductor placed in a magnetic field largest ?
Answer:
The force experienced by current carrying conductor placed in a magnetic field is largest when the current carrying conductor is placed in a direction perpendicular to that of magnetic field.
Question 15.
What change will you observe it you test soap with Litmus paper (red and blue)
Answer:
Soap is basic in nature and hence turns red litmus blue.
![]()
Question 16.
Define Rancidity ?
The oxidation of oils or fats in food resulting in a bad smell and bad taste is called rancidity.
III. Answer the following questions. ( 8 × 2 = 16 )
Question 17.
Define the term functional group identify the functional group present in the following.

It is a atom or group of atom or reactive part of compound which largely determine the chemical properties of compound
i) Methanol: Alcohol (-OH)
ii) Ethanoic acid : caboxylic acid (-COOH)
OR
What are saturated and unsaturated hydrocarbon? Write the structure of the simplest hydrocarbon ?
Answer:
- The hydrocarbon in which valency of carbon is saturated hydrocarbon.
- The valencies of all the atoms are satisfied by double or triple bonds between them are called unsaturated hydrocarbon.
- The simplest hydrocarbon is methane.

![]()
Question 18.
Draw the diagram of an electric circuit in which the resistors R1,R2, and R3, are connected in parallel including an ammeter and a voltmeter and mark the direction of the current.
Answer:
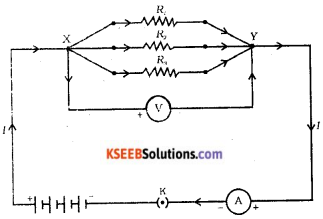
Question 19.
Name the part of the eye where image is formed by the eye lens. What is the nature of the image formed ? How is their sent to the brain ?
Answer:
The image of the object formed by the eye lens is at the retina of the eye. The image formed on the retina is real and inverted the image is sent to the brain with the help of the optic nerve.
Question 20.
How are the alveoli designed to maximise the exchange of gases ?
Answer:
There are millions of thin walled air sacs called alveoli in the lungs. The presence of these alveoli provides a large area for exchange of gases the availability of large surface area maximises the exchange of gases for example if all the alveoli from two human beings are unfolded they would give an area of about 80 square metres.
OR
How is the amount of urine produced in human regulated ?
Answer:
The amount of urine produced is regulated by reabsorption of water and some of the dissolved subtances into the blood through blood capillaries surrounded by the tabules of nephrons. The amount of urine produced depends upon how much excess water is present in the body and how much of dissolved salts are to be excreted.
Question 21.
Draw a diagram of testing the conductivity of a salt solution ?
Answer:
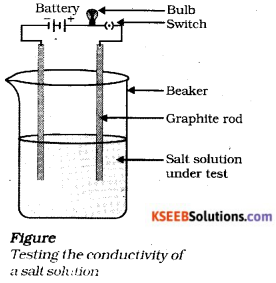
Question 22.
Distinguish between renewable and non-renewable sources of energy.
Answer:
Renewable source:
The sources of energy which can never be finished and are continuously produced in nature are known as renewable sources of energy.
Ex: wind, the sun, biogas, hydropower etc.
Non-renewable sources:
The sources of energy which are exahaustable and take lots of time to be formed again are known as non-renewable source of energy.
For ex: coal, natural gas petroleum etc.
Question 23.
Draw the diagram showing longitudinal section of a flower and lable the parts where pollination takes place.
Answer:
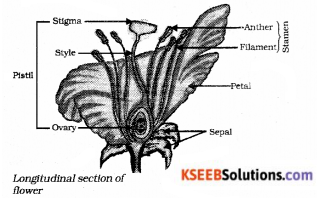
![]()
Question 24.
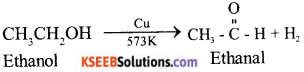
Describe two examples of different oxidations of ethanol. Name the products obtained in each case
Answer:
i) When ethanol is heated with copper at 573 k, Ethanol if is formed.
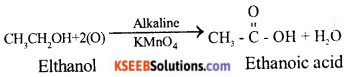
ii) When Ethanol is oxidised with alkaline
potassium permanganate solution, ethanoic acid is formed.
IV. Answer the following questions. ( 9 × 3 = 27 )
Question 25.
The atomic number of Na and Mg is 11 and 12 respectively and they belong to the same period.
a) Which one would have smaller atomic size.
b) Which one would be more electro positive.
c) To which group would each one belong
Answer:
a) Mg has smaller atomic size due to high nuclear charge (Na → 2, 8, 1; Mg → 2, 8, 2)
b) Na would be more electro positive because it can lose the valance electron more easily.
c) Na belongs to group 1 and Mg belong to group 2 as Na contains 1 and Mg contains 2 valence electron.
OR
How did Dobereiner classify the elements? Give one example of what were the limitations of this classification.
Answer:
He classified in group of three such that atomic No. of middle elements is average of first and third element.
Eg : Li(7), Na(23), K(39).
Limitations :
- Dobereiner could find only three triads, i,e, total of 9 elements only.
- However the total number of elements were more than that of those encompassed in Dobereiner’s triad.
- Thus Dobereiner could not classify most of the elements known at that time.
Question 26.
What is meant by sustainable management ? The environmentalist are insisting upon “Sustainable natural resource management” State its four advantages.
Answer:
Sustainable management is the management of natural resources which requires a long term perspective so that it lasts for generation to come and should not be exploited for short term gains.
Its four advantages are :
- Resources last for a longer duration
- It provides steady economic growth
- It helps in ecological conservation
- It reduces pollution.
Question 27.
What are marginal rays ? A 4 cm tall object is placed perpendicular to the principal axis of a convex lens of focal length 24 cm, the distance of the object from the lens is 16 cm find the position size and nature of the image formed using the lens formula.
Answer:
The ray which stricks the mirror surface near the periphery are called marginal rays for convex lens,
f = +24 cm, u= 16 cm
using lens formula

The image is formed at a distance of 48 cm from the optical focus of the lens on the same side of the object. This is indicated by the negative sign. Size of image is three times the size of object i.e., 12 cm.
Nature of image: Positive sign in the image height indicates that image is virtual and erect.
OR
Define the power of lens. What is the meaning of “the power of lens is 1 dioptre” if the power of a lens -2.0D, then what type of lens is that ? when an object is kept at infinity from this type of lens, what is the size of the image formed.
Answer:
The ability of a lens to converge or diverge the ray of light after refraction is called power (p) of the lens. It is defined as the reciprocal of the focal length.
i.e, P = \(\frac{1}{f}\)
SI unit of power of a lens is diopter. A lens of focal length 100 cm has a power of 1 dioptre i,e., 1 dioptre = 1 m
If the power of a lens is 2 D then the type of lens is concave lens.
The size of the image in this lens is highly diminished point sized.
![]()
Question 28.
Draw the diagram showing the sectional view of the human heart. Label the following parts.
i) Aorta
ii) Chamber of the heart that receives deoxygenated blood
Answer:
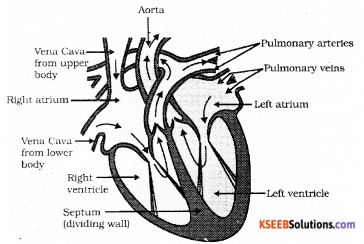
Question 29.
a) Why do ionic compounds have high melting points ?
b) Write the equation for the reaction of
i) Iron with steam
ii) Calcium and potassium with water.
Answer:
In the formation of ionic compounds, positive ions and negative ions precipitate. These are closely packed and the ionic compounds exist as crystalline solids. They have strong inter – ionic process of attraction and have high metling and boiling points.
i) 3Fe + 4H2O → FC2O4 + H2
ii) Ca + 2H2O → Ca(OH)2 + H2
2K + 2H2O → 2KOH + H2
OR
a) What is thermite reaction ?
b) Q is an element which is one in copper, zinc, aluminium and iron, its properties are as follows.
i) Q2 Q3 is found in nature but not affected by water.
ii) Two chlorides with formula QCl2 and QCl3 are formed by the metal. Identify the metal
Answer:
a) The displacement reaction which is. highly exothermic and the metal produced is in molten state is called thermite reaction.
i) Since the oxide of the metal is Q2Q3 its valence is 3. therefore out of the mentioned metal only aluminium and iron can be the metal with properties mentioned above.
ii) QCl2 and QCl2 must be iron as it shows variable valency of two and three. FeCl2 and FeCl3 are the chlorides.
Question 30.
Draw the ray diagram showing the image formation by a convex lens, when the object is kept beyond 2F, with the help of the diagram mention the nature of the image formed.
Answer:
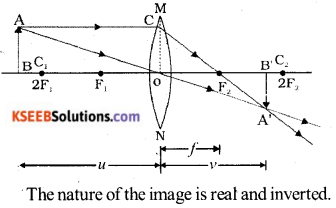
Question 31.
What is the function of receptors in our body? think of situations where receptors do not work properly what problemsare likely to arise ?
Answer:
Receptors are specialised called tissues, organ game and nerve ending which are able to pick up specific stimuli e.g gustatory receptors help in detecting taste and olfactory receptors help in sensing smell receptors provide sensory input about external and internal environment. Without them an animal will not be able to observe handle and taste food.
It will, not be aware of an approaching enemy. The animal may not be able to correct its positions and will fall down repeatedly. Therefore animal will not be able to perform the normal activities connected with the defective receptors.
OR
a) Name the hormone secreted by an endocrine gland during emergency.
b) Compare and contrast nervous and hormonal mechanism for control and co – ordination in animals ?
Answer:
a) Adrenalin hormone is secreted by the adrenal gland. It helps to regulate heartbeat, blood pressure, metabolism in the times of stress or emergency to cope up with the situation.
b) Nervous system:
- It consists of nerve impules between PNS, CNS and brain.
- The axone and dendrite transmit through a coordinated effort.
- The flow of information is rapid and the response is quick,
- Nerve impulses are not specific in their action.
- Effects are short level.
Hormonal system:
- It consists of endocrine system which secrete hormones directly into blood.
- The information is transmitted or transported through blood.
- The information travels slowly and the response is slow.
- Each hormone has specific action
- It has prolonged effects.
![]()
Question 32.
Write the chemical equation of the reaction in which the following changes have taken place with an example of each.
Answer:
i) Change in colour
Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag.
The solution will become blue in colour and shining silver metal will be deposited
ii) Change in temperature.
NaOH + HCl → NaCl + H2O + Heat.
The temperature will increase because heat is evolved.
iii) Formation of precipitate.
Pb(NO3)2(aq) +2KI(aq) → PbI2(s) +2KNO3(aq)
Yellow PPt
Yellow precipitate of Phi, will he formed.
Question 33.
State the principle of an electric generator. Write any two differences between electric motors and electric generators.
Answer:
An electric generator works on the principle of electromagnetic induction when, a closed coil is rotated in a uniform magnetic field with its axis perpendicular to the direction of the field. The magnetic field lines passing through the coil changes and induce potential differences. Hence a current is set up in it.
Electric motor:
- It converts electrical energy into mechanical energy,
- It works on the principle of magnetic field of electric current.
Electric generator:
- It converts mechanical energy into electrical energy.
- It works on the principle of electromagnetic induction.
V. Answer the following questions. ( 4 × 4 = 16 )
Question 34.
An electric geyser rated 1500 W, 250 V is connected to a 250 V line mains. Calculate the electric current draw n by it, energy consumed by it in 50 hours and energy consumed if each unit coast Rs. 6.00.
Answer:
Given P = 1500 WV = 250 V
i) Current drawn I = \(\frac{P}{V}=\frac{1500}{250}\) = 6A
ii) Electric energy consumed by gyser in
50 hours = Power × time
= 1500 × 50 = 75,000 wh
= 75 kwh
Since 1 kwh = 1 unit
75 kwh = 75 units.
∴Energy consumed = 75 units
iii) Cost of one unit = 6.00 Cost
75 units = 75 × 6.00
Rs. 450.00
![]()
Question 35.
i) Write the difference between homologous organs and analogous organs.
ii) Write the difference between the sex chromosomes of man and sex chromosomes of woman.
iii) Sex of a child is determined by the father how ?
Answer:
i) Difference between homologous analogous.
Homologous organs:
- Organs of different organisms have common origin.
- They have similar structure and perform different function.
- Ex. Forelimbs of frog and forelimbs of bird.
Analogous organs:
- Organs of different organisms have different origin they have different structure and perform similar function.
- Wings of bird and wings of bat.
ii) Woman have a perfect pair of sex chromosomes both called X. Man has a normal sized chromosome X and another short sized chromosome Y.
iii) A child who inherits X chromosome from her father will be a girl and child who inherits Y chromosome from his father will be a boy both the girl and the boy inherit only X chromosomes from the mother therefore sex of a child is determined by the father.
OR
a) What is meant by the term speciation. List four factors which could lead to speciation.
b) Name the two laws of inheritance postulated by Mendel.
Answer:
a) Speciation is the evolution of reproduction isolation among once interbreeding population factors which can lead to speciation are:
- Genetic drift : over generations, genetic drift may accumulate which lead to speciation.
- Natural selection : Natural selection which may give rise to speciation.
- Severe DNA change
- A variation may occur which does not allow sexual act between two groups.
b) i) The law of regeneration
ii) The law of independence assortment.
Question 36.
Why does the sun appear reddish early in the morning. Why does the sky appear dark instead of blue to an astronaut.
Answer:
During sunrise the light rays coming from the sun have to travel a greater distance in the earth’s atmosphere before reaching our eyes in this journey. The shorter waves of light are scattered out and only longer wavelengths are able to reach our eyes. Since blue colour has a shorter wave length and red colour has longer wave length the red colour is able to reach our eye after the atmospheric scattering of light, therefore the sun appears reddish early in the morning.
The sky appears dark instead of blue to an astronaut because there is no atmosphere in the outer space that can scatter the sunlight as the sunlight reaches the eyes of the astronauts. Without scattering the sky appears black to them.
![]()
Question 37.
a) How is plaster of paris prepared ? What reaction takes place when it sets to a hard mass.
Answer:
Plaster of paris is prepared by heating gypsum to a temperature of 100°C. The following reaction takes place :
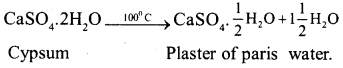
On mixing with water it retracts to a hard mass to form gypsum again i.e., the reverse reaction takes place.
b) Explain the general properties of bases
Answer:
- Bases possess a bitter taste
- Bases are slippery to touch.
- Bases turn red litmus blue
- Bases turn phenolpthalein solution pink
- Strong base like caustic soda (NaOH) caustic potash (KOH) are corrosive in nature and produce burning sensation on the skin
- Solution of bases in water conduct electricity.
- Bases react with acid, neutralise them.
- Producing salt and water.
![]()
VI. Answer the following question. ( 1 × 5 = 5 )
Question 38.
a) What are the advantages of sexual reproduction over asexual reproduction.
b) List any four reasons for adopting contraceptive method
c) If a woman is using copper T, will it help in protecting her from sexually transmitted diseases. Why ?
Answer:
a) Advantages of sexual reproduction.
- In sexual reproduction more variations are produced thus it ensures survival of species in a pollination.
- The new formed individual has characteristics of both the parents.
- Variations are more variable in sexual mode then in asexual one. This is because in asexual reproduction, DNA has to function inside the inherited cellular apparatus.
b) Four reasons for adopting contraceptive methods are
- To increase the gap between two children.
- To prevent unwanted pregnancy.
- To prevent transmission of STD’s
- To control populations growth.
c) If a woman is using copper – T it will not help in protecting her from sexually transmitted diseases. Copper – T prevents only implantation in the uterus.